质量管理体系:组织发展的工具还是额外负担?
- 作者: Zayunchkovskiy S.Y.1, Konovalov S.A.1, Zinchenko V.V.1, Sharova D.E.1, Ahkmad E.S.1, Vladzymyrskyy A.V.1
-
隶属关系:
- Moscow Center for Diagnostics and Telemedicine
- 期: 卷 4, 编号 3 (2023)
- 页面: 439-447
- 栏目: 致编辑的一封信
- URL: https://journals.rcsi.science/DD/article/view/254081
- DOI: https://doi.org/10.17816/DD514629
- ID: 254081
如何引用文章
详细
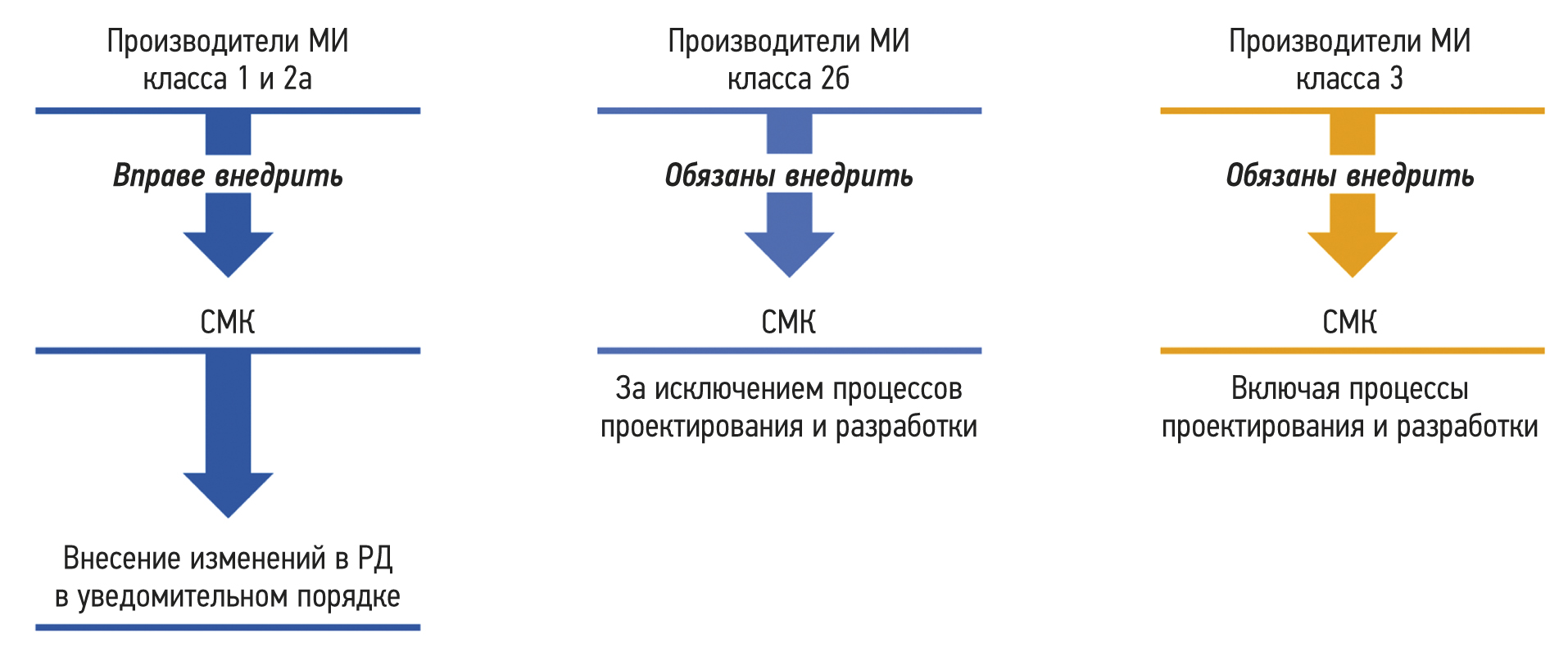
质量管理体系是组织的管理体系之一,它规定了组织活动中的一系列流程,旨在确保产品和服务的稳定质量。
随着全球工业生产的增长,有必要建立特殊的生产和管理系统,以确保企业能够根据消费者的要求随时满足制成品不断变化的客户价值,并使消费者本身感到满意。在这方面,人们开始关注组织内部在创造产品时实施的生产流程。就医疗器械生产而言,质量管理体系可定义为:与医疗产品质量有关的生产组织的协调领导和管理所需的组织结构、职能、程序、流程和资源。
该文章介绍质量管理体系的原则和管理流程。主要关注医疗器械质量管理体系的特殊性,包括作为医疗器械的软件质量管理体系的特殊性。本文指出质量管理体系成为确保组织可持续发展工具的条件。
作者简介
Sergey Yu. Zayunchkovskiy
Moscow Center for Diagnostics and Telemedicine
Email: ZayunchkovskijSY@zdrav.mos.ru
ORCID iD: 0009-0002-7463-7699
俄罗斯联邦, Moscow
Sergey A. Konovalov
Moscow Center for Diagnostics and Telemedicine
Email: KonovalovSA4@zdrav.mos.ru
ORCID iD: 0009-0003-0011-3371
俄罗斯联邦, Moscow
Viktoria V. Zinchenko
Moscow Center for Diagnostics and Telemedicine
编辑信件的主要联系方式.
Email: ZinchenkoVV1@zdrav.mos.ru
ORCID iD: 0000-0002-2307-725X
SPIN 代码: 4188-0635
俄罗斯联邦, Moscow
Daria E. Sharova
Moscow Center for Diagnostics and Telemedicine
Email: SharovaDE@zdrav.mos.ru
ORCID iD: 0000-0001-5792-3912
SPIN 代码: 1811-7595
俄罗斯联邦, Moscow
Ekaterina S. Ahkmad
Moscow Center for Diagnostics and Telemedicine
Email: AkhmadES@zdrav.mos.ru
ORCID iD: 0000-0002-8235-9361
SPIN 代码: 5891-4384
俄罗斯联邦, Moscow
Anton V. Vladzymyrskyy
Moscow Center for Diagnostics and Telemedicine
Email: VladzimirskijAV@zdrav.mos.ru
ORCID iD: 0000-0002-2990-7736
SPIN 代码: 3602-7120
MD, Dr. Sci. (Med.)
俄罗斯联邦, Moscow参考
- Mikhailov YuI. Process management in the quality management system of the enterprise. Discourse. 2017;(6):51–57. (In Russ).
- Zinovieva EV, Sapunova AV, Ivanov IV. Safety of circulation of medical devices at all stages of their life cycle. Public health. 2022;2(3):16–24. (In Russ). doi: l0.21045/2782-1676-202l-2-3-16-24
- Regulation (EU) 2017/745 of the European Parliament and of the Council of 5 April 2017 on medical devices, amending Directive 2001/83/EC, Regulation (EC) No 178/2002 and Regulation (EC) No 1223/2009 and repealing Council Directives 90/385/EEC and 93/42/EEC // Official J Eur Union. Available from: https://eur-lex.europa.eu/legal-content/EN/TXT/PDF/?uri=CELEX:32017R0745. Accessed: 15.07.2023.
- Lim K, Heo TY, Yun J. Trends in the approval and quality management of artificial intelligence medical devices in the Republic of Korea. Diagnostics (Basel). 2022;12(2):355. doi: 10.3390/diagnostics12020355
- Gusev AV, Vladzimirsky AV, Sharova DE, et al. Development of research and development in the field of artificial intelligence technologies for healthcare in the Russian Federation: Results of 2021. Digital Diagnostics. 2022;3(3):178–194. (In Russ). doi: 10.17816/DD107367
补充文件