Sarcopenia: modern approaches to solving diagnosis problems
- Authors: Smorchkova A.K.1, Petraikin A.V.1, Semenov D.S.1, Sharova D.E.1
-
Affiliations:
- Research and Practical Clinical Center for Diagnostics and Telemedicine Technologies
- Issue: Vol 3, No 3 (2022)
- Pages: 196-211
- Section: Reviews
- URL: https://journals.rcsi.science/DD/article/view/110721
- DOI: https://doi.org/10.17816/DD110721
- ID: 110721
Cite item
Abstract
Although sarcopenia is a relatively new diagnosis for medical statistics and the healthcare system, it represents a social and economic burden on the healthcare due to the large number of possible adverse outcomes such as increased risk of falls, physical disability, longer hospital stays, and increased mortality. No specialized medical treatment is available for sarcopenia; however, prevention and timely nonpharmacological treatment can reduce the risk of potential adverse effects. To establish the diagnosis of sarcopenia, it is necessary to confirm the decrease in not only muscle strength but also muscle mass. Instrumental diagnostics includes methods such as dual-energy X-ray absorptiometry and bioimpedance analysis. These methods can be supplemented by artificial intelligence algorithms for the automatic segmentation of muscle and fat tissue on computed tomography and magnetic resonance images, followed by calculation of the skeletal muscle index at the level of the L3 vertebra (L3SMI). Such software, when used in systems such as the Unified Radiological Information Service of the Unified Medical Information and Analytical System of Moscow, opens up opportunities for opportunistic screening. However, despite the recognition of CT and MRI as the “gold standard” by the European Working Group on Sarcopenia in Older People, there are no generally accepted L3SMI cut-off values for CT and MR diagnostics of sarcopenia. Furthermore, there is the problem of unifying the term “skeletal muscle index.” If these problems could be solved through further population studies, it will be possible to obtain a new method for the instrumental diagnosis of sarcopenia with its subsequent use for opportunistic screening.
Keywords
Full Text
##article.viewOnOriginalSite##About the authors
Anastasia K. Smorchkova
Research and Practical Clinical Center for Diagnostics and Telemedicine Technologies
Email: a.smorchkova@npcmr.ru
ORCID iD: 0000-0002-9766-3390
SPIN-code: 4345-8568
Scopus Author ID: 57213145638
Russian Federation, Moscow
Alexey V. Petraikin
Research and Practical Clinical Center for Diagnostics and Telemedicine Technologies
Email: alexeypetraikin@gmail.com
ORCID iD: 0000-0003-1694-4682
SPIN-code: 6193-1656
MD, Dr. Sci. (Med.)
Russian Federation, MoscowDmitry S. Semenov
Research and Practical Clinical Center for Diagnostics and Telemedicine Technologies
Email: d.semenov@npcmr.ru
ORCID iD: 0000-0002-4293-2514
SPIN-code: 2278-7290
Scopus Author ID: 57213154475
ResearcherId: P-5228-2017
Russian Federation, Moscow
Daria E. Sharova
Research and Practical Clinical Center for Diagnostics and Telemedicine Technologies
Author for correspondence.
Email: d.sharova@npcmr.ru
ORCID iD: 0000-0001-5792-3912
SPIN-code: 1811-7595
Russian Federation, Moscow
References
- Cruz-Jentoft AJ, Bahat G, Bauer J, et al. Sarcopenia: revised European consensus on definition and diagnosis. Age Ageing. 2019;48(1):16−31. doi: 10.1093/ageing/afy169
- Tkacheva ON, Kotovskaya YV, Runikhina NK, et al. Clinical guidelines on Frailty. Russ J Geriatric Med. 2020;(1):11−46. (In Russ). doi: 10.37586/2686-8636-1-2020-11-46
- Bischoff-Ferrari HA, Orav JE, Kanis JA, et al. Comparative performance of current definitions of sarcopenia against the prospective incidence of falls among community-dwelling seniors age 65 and older. Osteoporos Int. 2015;26(12):2793–2802 doi: 10.1007/s00198-015-3194-y
- da Silva Alexandre T, de Oliveira Duarte YA, Ferreira Santos JL, et al. Sarcopenia according to the European working group on sarcopenia in older people (EWGSOP) versus Dynapenia as a risk factor for disability in the elderly. J Nutr Health Aging. 2014;18(5):547−553. doi: 10.1007/s12603-014-0465-9
- Sousa AS, Guerra RS, Fonseca I, et al. Sarcopenia and length of hospital stay. Eur J Clin Nutr. 2016;70(5):595−601. doi: 10.1038/ejcn.2015.207
- Faulkner JA, Larkin LM, Claflin DR, Brooks SV. Age-related changes in the structure and function of skeletal muscles. Clin Exp Pharmacol Physiol. 2007;34(11):1091−1096. doi: 10.1111/j.1440-1681.2007.04752.x
- Shafiee G, Keshtkar A, Soltani A, et al. Prevalence of sarcopenia in the world: a systematic review and meta-analysis of general population studies. J Diabetes Metab Disord. 2017;16:21. doi: 10.1186/s40200-017-0302-x
- Petermann-Rocha F, Balntzi V, Gray SR, et al. Global prevalence of sarcopenia and severe sarcopenia: a systematic review and meta-analysis. J Cachexia Sarcopenia Muscle. 2022;13(1):86−99. doi: 10.1002/jcsm.12783
- Safonova YA, Zotkin EG. Sarcopenia in older patients with osteoarthritis of large joints. Sci Pract Rheumatol. 2019;57(2):154−159. (In Russ). doi: 10.14412/1995-4484-2019-154-159
- Tsekoura M, Kastrinis A, Katsoulaki M, et al. Sarcopenia and its impact on quality of life. Adv Exp Med Biol. 2017;987:213−218. doi: 10.1007/978-3-319-57379-3_19
- Sepúlveda-Loyola W, Osadnik C, Phu S, et al. Diagnosis, prevalence, and clinical impact of sarcopenia in COPD: a systematic review and meta-analysis. J Cachexia Sarcopenia Muscle. 2020;11(5):1164−1176. doi: 10.1002/jcsm.12600
- Nipp RD, Fuchs G, El-Jawahri A, et al. Sarcopenia is associated with quality of life and depression in patients with advanced cancer. Oncologist. 2018;23(1):97−104. doi: 10.1634/theoncologist.2017-0255
- Beaudart C, Biver E, Reginster JY, et al. Development of a self-administrated quality of life questionnaire for sarcopenia in elderly subjects: the SarQoL. Age Ageing. 2015;44(6):960−966. doi: 10.1093/ageing/afv133
- Geerinck A, Bruyère O, Locquet M, et al. Evaluation of the responsiveness of the SarQoL questionnaire, a patient-reported outcome measure specific to sarcopenia. Adv Ther. 2018;35(11):1842−1858. doi: 10.1007/s12325-018-0820-z
- Geerinck A, Locquet M, Bruyère O, et al. Evaluating quality of life in frailty: applicability and clinimetric properties of the SarQoL questionnaire. J Cachexia Sarcopenia Muscle. 2021;12(2):319−330. doi: 10.1002/jcsm.12687
- Witham MD, Heslop P, Dodds RM, et al. Performance of the SarQoL quality of life tool in a UK population of older people with probable sarcopenia and implications for use in clinical trials: findings from the SarcNet registry. BMC Geriatr. 2022;22(1):368. doi: 10.1186/s12877-022-03077-5
- Russian translation and validation of SarQoL ― quality of life questionnaire for patients with sarcopenia. Sci Pract Rheumatol. 2019;57(1):38−45. (In Russ). doi: 10.14412/1995-4484-2019-38-45
- Gani F, Buettner S, Margonis GA, et al. Sarcopenia predicts costs among patients undergoing major abdominal operations. Surgery. 2016;160(5):1162−1171. doi: 10.1016/j.surg.2016.05.002
- Bruyère O, Beaudart C, Ethgen O, et al. The health economics burden of sarcopenia: a systematic review. Maturitas. 2019;119:61−69. doi: 10.1016/j.maturitas.2018.11.003
- Peterson MD, Rhea MR, Sen A, Gordon PM. Resistance exercise for muscular strength in older adults: a meta-analysis. Ageing Res Rev. 2010;9(3):226−237. doi: 10.1016/j.arr.2010.03.004
- McKendry J, Currier BS, Lim C, et al. Nutritional supplements to support resistance exercise in countering the sarcopenia of aging. Nutrients. 2020;12(7):2057. doi: 10.3390/nu12072057
- Robinson SM, Reginster JY, Rizzoli R, et al. Does nutrition play a role in the prevention and management of sarcopenia? Clin Nutr. 2018;37(4):1121−1132. doi: 10.1016/j.clnu.2017.08.016
- Lozano-Montoya I, Correa-Pérez A, Abraha I, et al. Nonpharmacological interventions to treat physical frailty and sarcopenia in older patients: a systematic overview ― the SENATOR Project ONTOP Series. Clin Interv Aging. 2017;12:721−740. doi: 10.2147/CIA.S132496
- Lappe JM, Binkley N. Vitamin D and sarcopenia/falls. J Clin Densitom. 2015;18(4):478−482. doi: 10.1016/j.jocd.2015.04.015
- Rooks D, Roubenoff R. Development of pharmacotherapies for the treatment of sarcopenia. J Frailty Aging. 2019;8(3):120−130. doi: 10.14283/jfa.2019.11
- Morley JE, Abbatecola AM, Argiles JM, et al. Sarcopenia with limited mobility: an international consensus. J Am Med Dir Assoc. 2011;12(6):403−409. doi: 10.1016/j.jamda.2011.04.014
- Malmstrom TK, Miller DK, Simonsick EM, et al. SARC-F: a symptom score to predict persons with sarcopenia at risk for poor functional outcomes. J Cachexia Sarcopenia Muscle. 2016;7(1):28−36. doi: 10.1002/jcsm.12048
- Bahat G, Yilmaz O, Kiliç C, et al. Performance of SARC-F in regard to sarcopenia definitions, muscle mass and functional measures. J Nutr Health Aging. 2018;22(8):898−903. doi: 10.1007/s12603-018-1067-8
- Porto JM, Nakaishi AP, Cangussu-Oliveira LM, et al. Relationship between grip strength and global muscle strength in community-dwelling older people. Arch Gerontol Geriatr. 2019;82:273−278. doi: 10.1016/j.archger.2019.03.005
- Maggio M, Ceda GP, Ticinesi A, et al. Instrumental and non-instrumental evaluation of 4-meter walking speed in older individuals. PLoS One. 2016;11(4):e0153583. doi: 10.1371/journal.pone.0153583
- Podsiadlo D, Richardson S. The timed “Up & go”: a test of basic functional mobility for frail elderly persons. J Am Geriatr Soc. 1991;39(2):142−148. doi: 10.1111/j.1532-5415.1991.tb01616.x
- Beaudart C, McCloskey E, Bruyère O, et al. Sarcopenia in daily practice: assessment and management. BMC Geriatr. 2016;16(1):170. doi: 10.1186/s12877-016-0349-4
- Stringer HJ, Wilson D. The role of ultrasound as a diagnostic tool for sarcopenia. J Frailty Aging. 2018;7(4):258−261. doi: 10.14283/jfa.2018.24
- Petraikin AV, Smoliarchuk MY, Petryaykin FA, et al. Assessment the accuracy of densitometry measurements using DMA PP2 Phantom. Traumatol Orthopedics Russ. 2019;25(3):124−134. (In Russ). doi: 10.21823/2311-2905-2019-25-3-124-134
- Shen W, Punyanitya M, Wang Z, et al. Total body skeletal muscle and adipose tissue volumes: estimation from a single abdominal cross-sectional image. J Appl Physiol (1985). 2004;97(6):2333−2338. doi: 10.1152/japplphysiol.00744.2004
- Mourtzakis M, Prado CM, Lieffers JR, et al. A practical and precise approach to quantification of body composition in cancer patients using computed tomography images acquired during routine care. Appl Physiol Nutr Metab. 2008;33(5):997−1006. doi: 10.1139/H08-075
- Kim EY, Kim YS, Park I, et al. Prognostic significance of CT-determined sarcopenia in patients with small-cell lung cancer. J Thorac Oncol. 2015;10(12):1795−1799. doi: 10.1097/JTO.0000000000000690
- Baracos V, Kazemi-Bajestani SM. Clinical outcomes related to muscle mass in humans with cancer and catabolic illnesses. Int J Biochem Cell Biol. 2013;45(10):2302−2308. doi: 10.1016/j.biocel.2013.06.016
- Franceschi C, Garagnani P, Morsiani C, et al. The continuum of aging and age-related diseases: common mechanisms but different rates. Front Med (Lausanne). 2018;5:61. doi: 10.3389/fmed.2018.00061
- Ferrucci L, Fabbri E. Inflammageing: chronic inflammation in ageing, cardiovascular disease, and frailty. Nat Rev Cardiol. 2018;15(9):505−522. doi: 10.1038/s41569-018-0064-2
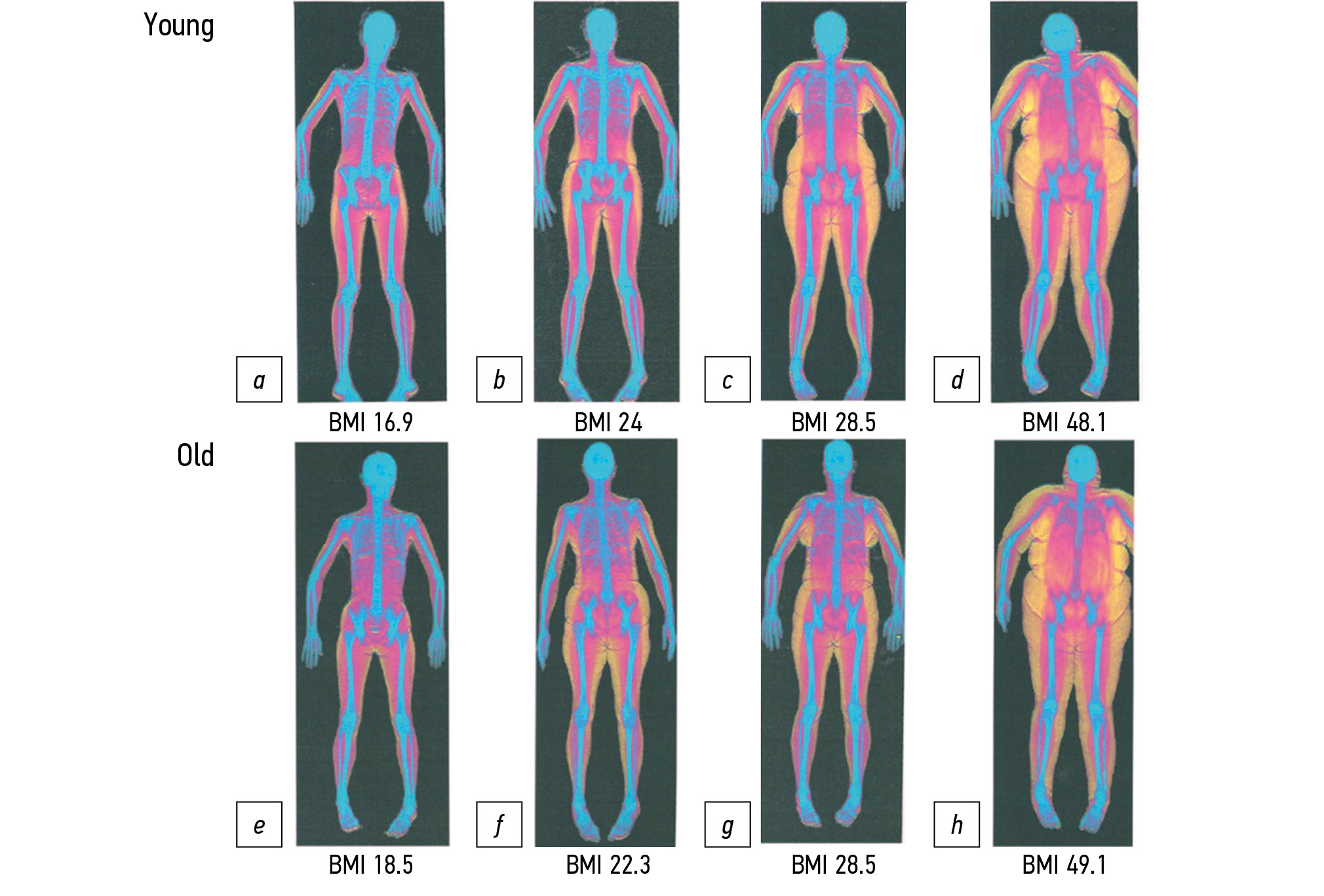
- Zamboni M, Rubele S, Rossi AP. Sarcopenia and obesity. Curr Opin Clin Nutr Metab Care. 2019;22(1):13−19. doi: 10.1097/MCO.0000000000000519
- Batsis JA, Villareal DT. Sarcopenic obesity in older adults: etiology, epidemiology and treatment strategies. Nat Rev Endocrinol. 2018;14(9):513−537. doi: 10.1038/s41574-018-0062-9
- Tomlinson DJ, Erskine RM, Winwood K, et al. Obesity decreases both whole muscle and fascicle strength in young females but only exacerbates the aging-related whole muscle level asthenia Physiol Rep. 2014;2(6):e12030. doi: 10.14814/phy2.12030
- Kim KM, Jang HC, Lim S. Differences among skeletal muscle mass indices derived from height-, weight-, and body mass index-adjusted models in assessing sarcopenia. Korean J Intern Med. 2016;31(4):643−650. doi: 10.3904/kjim.2016.015
- Newman AB, Kupelian V, Visser M, et al. Sarcopenia: alternative definitions and associations with lower extremity function. J Am Geriatr Soc. 2003;51(11):1602−1609. doi: 10.1046/j.1532-5415.2003.51534.x
- Ha J, Park T, Kim HK, et al. Development of a fully automatic deep learning system for L3 selection and body composition assessment on computed tomography. Sci Rep. 2021;11(1):21656. doi: 10.1038/s41598-021-00161-5
- Prado CM, Lieffers JR, McCargar LJ, et al. Prevalence and clinical implications of sarcopenic obesity in patients with solid tumours of the respiratory and gastrointestinal tracts: a population-based study. Lancet Oncol. 2008;9(7):629−635. doi: 10.1016/S1470-2045(08)70153-0
- Martin L, Birdsell L, Macdonald N, et al. Cancer cachexia in the age of obesity: skeletal muscle depletion is a powerful prognostic factor, independent of body mass index. J Clin Oncol. 2013;31(12):1539−1547. doi: 10.1200/JCO.2012.45.2722
- Popuri K, Cobzas D, Esfandiari N, et al. Body composition assessment in axial ct images using fem-based automatic segmentation of skeletal muscle. IEEE Trans Med Imaging. 2016;35(2):512−520. doi: 10.1109/TMI.2015.2479252
- Park J, Gil JR, Shin Y, et al. Reliable and robust method for abdominal muscle mass quantification using CT/MRI: an explorative study in healthy subjects. PLoS One. 2019;14(9):e0222042. doi: 10.1371/journal.pone.0222042
- Fedorov A, Beichel R, Kalpathy-Cramer J, et al. 3D Slicer as an image computing platform for the quantitative imaging network. Magn Reson Imaging. 2012;30(9):1323−1341. doi: 10.1016/j.mri.2012.05.001
- Burns JE, Yao J, Chalhoub D, et al. A machine learning algorithm to estimate sarcopenia on abdominal CT. Acad Radiol. 2020;27(3):311−320. doi: 10.1016/j.acra.2019.03.011
- Blanc-Durand P, Schiratti JB, Schutte K, et al. Abdominal musculature segmentation and surface prediction from CT using deep learning for sarcopenia assessment. Diagn Interv Imaging. 2020;101(12):789−794. doi: 10.1016/j.diii.2020.04.011
- Graffy PM, Liu J, Pickhardt PJ, et al. Deep learning-based muscle segmentation and quantification at abdominal CT: application to a longitudinal adult screening cohort for sarcopenia assessment. Br J Radiol. 2019;92(1100):20190327. doi: 10.1259/bjr.20190327
- Ackermans LL, Volmer L, Wee L, et al. Deep learning automated segmentation for muscle and adipose tissue from abdominal computed tomography in polytrauma patients. Sensors (Basel). 2021;21(6):2083. doi: 10.3390/s21062083
- Ronneberger O, Fischer P, Brox T. U-Net: convolutional networks for biomedical image segmentation. Lecture Notes Computer Sci. 2015:234−241. doi: 10.1007/978-3-319-24574-4_28
- Shelhamer E, Long J, Darrell T. Fully convolutional networks for semantic segmentation. IEEE Trans Pattern Anal Mach Intell. 2017;39(4):640−651. doi: 10.1109/tpami.2016.2572683
- Kanavati F, Islam S, Arain Z, et al. Fully-automated deep learning slice-based muscle estimation from CT images for sarcopenia assessment. Clin Radiol. 2022;77(5):e363−e371. doi: 10.1016/j.crad.2022.01.036
- Kim DW, Kim KW, Ko Y, et al. Assessment of myosteatosis on computed tomography by automatic generation of a muscle quality map using a web-based toolkit: feasibility study. JMIR Med Inform. 2020;8(10):e23049. doi: 10.2196/23049
- Dong X, Dan X, Yawen A, et al. Identifying sarcopenia in advanced non-small cell lung cancer patients using skeletal muscle CT radiomics and machine learning. Thorac Cancer. 2020;11(9):2650−2659. doi: 10.1111/1759-7714.13598
- Graffy PM, Liu J, Pickhardt PJ, et al. Deep learning-based muscle segmentation and quantification at abdominal CT: Application to a longitudinal adult screening cohort for sarcopenia assessment. Br J Radiol. 2019;92(1100):20190327. doi: 10.1259/bjr.20190327
- Petraikin AV, Artyukova ZR, Nizovtsova LA, et al. Analysis of the effectiveness of implementing screening of osteoporosis. Health Care Manager. 2021;(2):31−39. (In Russ). doi: 10.21045/1811-0185-2021-2-31-39
- Morozov SP, Vladzymyrsky AV, Ledikhova NV, et al. Moscow experiment on Computer Vision in radiology: Involvement and participation of Radiologists. Doctor Inform Tech. 2020;(4):14−23. (In Russ). doi: 10.37690/1811-0193-2020-4-14-23
- Senyukova OV, Pyatkovskiy SA, Petraikin AV, et al. Automatic segmentation of muscle and adipose tissue on CT images for assessing human body composition and diagnosing sarcopenia. In: Conference “Information Technologies for personalized medicine” with a block of the summer school for young scientists, November 4, 2021: collection of abstracts. Moscow; 2021. P. 41. (In Russ). doi: 10.14341/cbaipm-2021-41
Supplementary files