Новые методы визуализации атеросклероза
- Авторы: Syed M.B.1, Fletcher A.J.1, Forsythe R.O.1, Kaczynski J.1, Newby D.E.1, Dweck M.R.1, R. van Beek E.J.1
-
Учреждения:
- University of Edinburgh
- Выпуск: Том 2, № 3 (2021)
- Страницы: 386-409
- Раздел: Обзоры
- URL: https://journals.rcsi.science/DD/article/view/71633
- DOI: https://doi.org/10.17816/DD71633
- ID: 71633
Цитировать
Полный текст
Аннотация
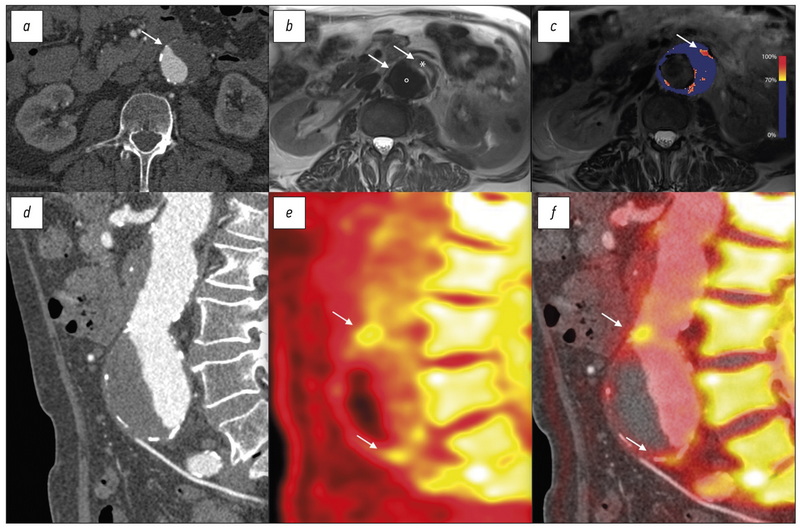
Атеросклероз ― это хроническое иммуномодулируемое заболевание, которое поражает артерии и приводит к значительному бремени болезней во всём мире. Традиционные методы визуализации сосредоточены на морфологических особенностях атеросклероза, таких как степень стеноза, вызванного поражением сосудов. С появлением современных компьютерных, магнитно-резонансных и позитронно-эмиссионных томографов повысились скорость получения изображений и пространственное разрешение, что обеспечивает широкие возможности их применения в клинической практике. Комплексная визуализация способствует более точному прогнозированию сердечно- сосудистого риска и даёт развёрнутую информацию о состоянии и метаболических процессах в стенках сосудов. Конкретные этапы развития болезни можно выявить с помощью биологических индикаторов и «умных» контрастных веществ. Эти подходы позволяют врачам оценить метаболическое состояние атеросклеротической бляшки.
В обзоре представлены данные о современных методах визуализации атеросклероза, применение которых поможет выявить основные морфологические характеристики заболевания.
Данная публикация является перепечатанной версией статьи с переводом оригинала [Syed MB, Fletcher AJ, Forsythe RO, Kaczynski J, Newby DE, Dweck MR, van Beek EJ. Emerging techniques in atherosclerosis imaging. Br J Radiol. 2019;92(1103):20180309. doi: 10.1259/bjr.20180309] на русский язык.
Полный текст
Открыть статью на сайте журналаОб авторах
Maaz B.J. Syed
University of Edinburgh
Email: maaz.syed@ed.ac.uk
ORCID iD: 0000-0003-0530-8306
Великобритания, Эдинбург
Alexander J. Fletcher
University of Edinburgh
Email: maaz.syed@ed.ac.uk
ORCID iD: 0000-0001-9984-8391
Великобритания, Эдинбург
Rachael O. Forsythe
University of Edinburgh
Email: maaz.syed@ed.ac.uk
ORCID iD: 0000-0002-3311-9599
Великобритания, Эдинбург
Jakub Kaczynski
University of Edinburgh
Email: maaz.syed@ed.ac.uk
ORCID iD: 0000-0002-3005-6860
Великобритания, Эдинбург
David E. Newby
University of Edinburgh
Email: maaz.syed@ed.ac.uk
ORCID iD: 0000-0001-7971-4628
Великобритания, Эдинбург
Marc R. Dweck
University of Edinburgh
Email: maaz.syed@ed.ac.uk
ORCID iD: 0000-0001-9847-5917
Великобритания, Эдинбург
Edwin J. R. van Beek
University of Edinburgh
Автор, ответственный за переписку.
Email: maaz.syed@ed.ac.uk
ORCID iD: 0000-0002-2777-5071
Великобритания, Эдинбург
49 Little France Crescent, Edinburgh EH16 4SB
Список литературы
- Herrington W., Lacey B., Sherliker P., et al. Epidemiology of atherosclerosis and the potential to reduce the global burden of atherothrombotic disease//Circ Res. 2016. Vol. 118, N 4. Р. 535–546. doi: 10.1161/CIRCRESAHA.115.307611
- Puchner S.B., Liu T., Mayrhofer T., et al. High-Risk plaque detected on coronary ct angiography predicts acute coronary syndromes independent of significant stenosis in acute chest pain//J Am Coll Cardiol. 2014. Vol. 64, N 7. Р. 684–692. doi: 10.1016/j.jacc.2014.05.039
- Teague H.L., Ahlman M.A., Alavi A., et al. Unraveling vascular inflammation: from immunology to imaging//J Am Coll Cardiol. 2017. Vol. 70, N 11. Р. 1403–1412. doi: 10.1016/j.jacc.2017.07.750
- Davignon J., Ganz P. Role of endothelial dysfunction in atherosclerosis//Circulation 2004. Vol. 109, N 23, Suppl 1. Р. III-27-20. doi: 10.1161/01.CIR.0000131515.03336.f8
- Pirillo A., Bonacina F., Norata G.D., Catapano A.L. The interplay of lipids, lipoproteins, and immunity in atherosclerosis//Curr Atheroscler Rep. 2018. Vol. 20, N 3. Р. 12. doi: 10.1007/s11883-018-0715-0
- Mullick A.E., Soldau K., Kiosses W.B., et al. Increased endothelial expression of Toll-like receptor 2 at sites of disturbed blood flow exacerbates early atherogenic events//J Exp Med. 2008. Vol. 205, N 2. Р. 373–383. doi: 10.1084/jem.20071096
- Newby A.C. Metalloproteinase production from macrophages — a perfect storm leading to atherosclerotic plaque rupture and myocardial infarction//Exp Physiol. 2016. Vol. 101, N 11. Р. 1327–1337. doi: 10.1113/EP085567
- Stary H.C., Chandler A.B., Dinsmore R.E., et al. A definition of advanced types of atherosclerotic lesions and a histological classification of atherosclerosis: a report from the Committee on vascular lesions of the Council on arteriosclerosis, American heart association//Circulation. 1995. Vol. 92, N 5. Р. 1355–1374. doi: 10.1161/01.cir.92.5.1355
- Virmani R., Burke A.P., Kolodgie F.D., Farb A. Pathology of the thin-cap fibroatheroma//J Intervent Cardiol. 2003. Vol. 16, N 3. Р. 267–272. doi: 10.1034/j.1600-0854.2003.8042.x
- Sluimer J.C., Gasc J.M., van Wanroij J.L., et al. Hypoxia, hypoxia-inducible transcription factor, and macrophages in human atherosclerotic plaques are correlated with intraplaque angiogenesis//J Am Coll Cardiol. 2008. Vol. 51, N 13. Р. 1258–1265. doi: 10.1016/j.jacc.2007.12.025
- Fuery M.A., Liang L., Kaplan F.S., Mohler E.R. Vascular ossification: pathology, mechanisms, and clinical implications//Bone. 2018. Vol. 109. Р. 28–34. doi: 10.1016/j.bone.2017.07.006
- Hirsch D., Azoury R., Sarig S., Kruth H.S. Colocalization of cholesterol and hydroxyapatite in human atherosclerotic lesions//Calcif Tissue Int. 1993. Vol. 52, N 2. Р. 94–98. doi: 10.1007/BF00308315
- Virmani R., Burke A.P., Kolodgie F.D., Farb A. Pathology of the thin-cap fibroatheroma: a type of vulnerable plaque//J Interv Cardiol. 2003. Vol. 16, N 3. Р. 267–272. doi: 10.1034/j.1600-0854.2003.8042.x
- Emberson J., Lees K.R., Lyden P., et al. Effect of treatment delay, age, and stroke severity on the effects of intravenous thrombolysis with alteplase for acute ischaemic stroke: a meta-analysis of individual patient data from randomised trials//Lancet. 2014. Vol. 384, N 9958. Р. 1929–1935. doi: 10.1016/S0140-6736(14)60584-5
- Jang I.K., Tearney G.J., MacNeill B., et al. In vivo characterization of coronary atherosclerotic plaque by use of optical coherence tomography//Circulation. 2005. Vol. 111, N 12. Р. 1551–1555. doi: 10.1161/01.CIR.0000159354.43778.69
- Tearney G.J., Yabushita H., Houser S.L., et al. Quantification of macrophage content in atherosclerotic plaques by optical coherence tomography//Circulation. 2003. Vol. 107, N 1. Р. 113–119. doi: 10.1161/01.CIR.0000044384.41037.43
- Bouma B.E., Tearney G.J., Yabushita H., et al. Evaluation of intracoronary stenting by intravascular optical coherence tomography//Heart. 2003. Vol. 89, N 3. Р. 317–320. doi: 10.1136/heart.89.3.317
- Jang I.K., Bouma B.E., Kang D.H., et al. Visualization of coronary atherosclerotic plaques in patients using optical coherence tomography: comparison with intravascular ultrasound//J Am Coll Cardiol. 2002. Vol. 39, N 4. Р. 604–609. doi: 10.1016/S0735-1097(01)01799-5
- Dohad S., Zhu A., Krishnan S., et al. Optical coherence tomography guided carotid artery stent procedure: technique and potential applications//Catheter Cardiovasc Interv. 2018. Vol. 91, N 3. Р. 521–530. doi: 10.1002/ccd.27344
- Schwindt A.G., Bennett J.G., Crowder W.H., et al. Lower extremity revascularization using optical coherence tomography ― guided directional atherectomy: final results of the evaluati on of the pantheris optical coherence tomography imaging atherectomy system for use in the peripheral vasculature (VISION) study//J Endovasc Ther. 2017. Vol. 24, N 3. Р. 355–366. doi: 10.1177/1526602817701720
- Waxman S., Dixon S.R., L'Allier P., et al. In vivo validation of a Catheter-Based near-infrared spectroscopy system for detection of lipid core coronary plaques//JACC Cardiovascular Imaging. 2009. Vol. 2, N 7. Р. 858–868. doi: 10.1016/j.jcmg.2009.05.001
- Voros S., Rinehart S., Qian Z., et al. Prospective validation of standardized, 3-dimensional, quantitative coronary computed tomographic plaque measurements using radiofrequency Backscatter intravascular ultrasound as reference standard in intermediate coronary arterial lesions: results from the Atlanta (assessment of tissue characteristics, lesion morphology, and hemodynamics by angiography with fractional flow reserve, intravascular ultrasound and virtual histology, and noninvasive computed tomography in atherosclerotic plaques) I study//JACC Cardiovasc Interv. 2011. Vol. 4, N 2. Р. 198–208. doi: 10.1016/j.jcin.2010.10.008
- Broersen A., de Graaf M.A., Eggermont J., et al. Enhanced characterization of calcified areas in intravascular ultrasound virtual histology images by quantification of the acoustic shadow: validation against computed tomography coronary angiography//Int J Cardiovasc Imaging 2016. Vol. 32, N 4. Р. 543–552. doi: 10.1007/s10554-015-0820-x
- Brugaletta S., Garcia-Garcia H.M., Serruys P.W., et al. Nirs and IVUS for characterization of atherosclerosis in patients undergoing coronary angiography//JACC Cardiovascular Imaging. 2011. Vol. 4, N 6. Р. 647–655. doi: 10.1016/j.jcmg.2011.03.013
- Štěchovský C., Hájek P., Horváth M., et al. Near-Infrared spectroscopy combined with intravascular ultrasound in carotid arteries//Int J Cardiovasc Imaging. 2016. Vol. 32, N 1. Р. 181–188. doi: 10.1007/s10554-015-0687-x
- Yin D., Matsumura M., Rundback J., et al. Comparison of plaque morphology between peripheral and coronary artery disease (from the clarity and ADAPT-DES IVUS substudies//Coron Artery Dis. 2017. Vol. 28, N 5. Р. 369–375. doi: 10.1097/MCA.0000000000000469
- Mitchell C., Korcarz C.E., Gepner A.D., et al. Ultrasound carotid plaque features. cardiovascular disease risk factors and events: the multi-ethnic study of atherosclerosis//Atherosclerosis. 2018. Vol. 276. Р. 195–202. doi: 10.1016/j.atherosclerosis.2018.06.005
- Lal B.K., Hobson R.W., Pappas P.J., et al. Pixel distribution analysis of B-mode ultrasound scan images predicts histologic features of atherosclerotic carotid plaques//J Vasc Surg. 2002. Vol. 35, N 6. Р. 1210–1217. doi: 10.1067/mva.2002.122888
- Finn A.V., Kolodgie F.D., Virmani R. Correlation between carotid intimal/medial thickness and atherosclerosis: a point of view from pathology//Arterioscler Thromb Vasc Biol. 2010. Vol. 30, N 2. Р. 177–181. doi: 10.1161/ATVBAHA.108.173609
- Stein J.H., Korcarz C.E., Hurst R.T., et al. Use of carotid ultrasound to identify subclinical vascular disease and evaluate cardiovascular disease risk: a consensus statement from the American Society of echocardiography carotid intima-media thickness Task force endorsed by the Society for vascular medicine//J Am Soc Echocardiogr. 2008. Vol. 21, N 2. Р. 93–111. doi: 10.1016/j.echo.2007.11.011
- Huang R., Abdelmoneim S.S., Ball C.A., et al. Detection of carotid atherosclerotic plaque neovascularization using contrast enhanced ultrasound: a systematic review and meta-analysis of diagnostic accuracy studies//J Am Soc Echocardiogr. 2016. Vol. 29, N 6. Р. 491–502. doi: 10.1016/j.echo.2016.02.012
- Mirza T.A., Karthikesalingam A., Jackson D., et al. Duplex ultrasound and contrast-enhanced ultrasound versus computed tomography for the detection of endoleak after EVAR: systematic review and bivariate meta-analysis//Eur J Vasc Endovasc Surg. 2010. Vol. 39, N 4. Р. 418–428. doi: 10.1016/j.ejvs.2010.01.001
- Moneta G.L., Edwards J.M., Chitwood R.W., et al. Correlation of North American Symptomatic Carotid Endarterectomy Trial (NASCET) angiographic definition of 70% to 99% internal carotid artery stenosis with duplex scanning//J Vasc Surg. 1993. Vol. 17, N 1. Р. 152–159. doi: 10.1016/0741-5214(93)90019-I
- Josephson S.A., Bryant S.O., Mak H.K., et al. Evaluation of carotid stenosis using CT angiography in the initial evaluation of stroke and TIA//Neurology. 2004. Vol. 63, N 3. Р. 457–460. doi: 10.1212/01.WNL.0000135154.53953.2C
- Maurovich-Horvat P., Ferencik M., Voros S., et al. Comprehensive plaque assessment by coronary CT angiography//Nat Rev Cardiol. 2014. Vol. 11, N 7. Р. 390–402. doi: 10.1038/nrcardio.2014.60
- Divakaran S., Cheezum M.K., Hulten E.A., et al. Use of cardiac CT and calcium scoring for detecting coronary plaque: implications on prognosis and patient management//Br J Radiol. 2015. Vol. 88, N 1046. Р. 20140594. doi: 10.1259/bjr.20140594
- Motoyama S., Ito H., Sarai M., et al. Plaque characterization by coronary computed tomography angiography and the likelihood of acute coronary events in mid-term follow-up//J Am Coll Cardiol. 2015. Vol. 66, N 4. Р. 337–346. doi: 10.1016/j.jacc.2015.05.069
- Maurovich-Horvat P., Hoffmann U., Vorpahl M., et al. The Napkin-Ring sign: CT signature of high-risk coronary plaques?//JACC Cardiovasc Imaging. 2010. Vol. 3, N 4. Р. 440–444. doi: 10.1016/j.jcmg.2010.02.003
- Fathi A., Weir-McCall J.R., Struthers A.D., et al. Effects of contrast administration on cardiac MRI volumetric, flow and pulse wave velocity quantification using manual and software-based analysis//Br J Radiol. 2018. Vol. 91, N 1084. Р. 20170717. doi: 10.1259/bjr.20170717
- Akçakaya M., Basha T.A., Chan R.H., et al. Accelerated isotropic sub-millimeter whole-heart coronary MRI: compressed sensing versus parallel imaging//Magn Reson Med. 2014. Vol. 71, N 2. Р. 815–822. doi: 10.1002/mrm.24683
- Kim W.Y., Danias P.G., Stuber M., et al. Coronary magnetic resonance angiography for the detection of coronary stenoses//N Engl J Med. 2001. Vol. 345, N 26. Р. 1863–1869. doi: 10.1056/NEJMoa010866
- Dweck M.R., Williams M.C., Moss A.J., et al. Computed tomography and cardiac magnetic resonance in ischemic heart disease//J Am Coll Cardiol. 2016. Vol. 68, N 20. Р. 2201–2216. doi: 10.1016/j.jacc.2016.08.047
- Hatsukami T.S., Ross R., Polissar N.L., Yuan C. Visualization of fibrous cap thickness and rupture in human atherosclerotic carotid plaque in vivo with high-resolution magnetic resonance imaging//Circulation. 2000. Vol. 102, N 9. Р. 959–964. doi: 10.1161/01.CIR.102.9.959
- Kerwin W.S., Zhao X., Yuan C., et al. Contrast-Enhanced MRI of carotid atherosclerosis: dependence on contrast agent//J Magn Reson Imaging. 2009. Vol. 30, N 1. Р. 35–40. doi: 10.1002/jmri.21826
- Chu B., Kampschulte A., Ferguson M.S., et al. Hemorrhage in the atherosclerotic carotid plaque: a high-resolution MRI study//Stroke. 2004. Vol. 35, N 5. Р. 1079–1084. doi: 10.1161/01.STR.0000125856.25309.86
- Saam T., Hetterich H., Hoffmann V., et al. Meta-Analysis and systematic review of the predictive value of carotid plaque hemorrhage on cerebrovascular events by magnetic resonance imaging//J Am Coll Cardiol. 2013. Vol. 62, N 12. Р. 1081–1091. doi: 10.1016/j.jacc.2013.06.015
- Kooi M.E., Cappendijk V.C., Cleutjens K.B., et al. Accumulation of ultrasmall superparamagnetic particles of iron oxide in human atherosclerotic plaques can be detected by in vivo magnetic resonance imaging//Circulation. 2003. Vol. 107, N 19. Р. 2453–2458. doi: 10.1161/01.CIR.0000068315.98705.CC
- Richards J.M., Semple S.I., MacGillivray T.J., et al. Abdominal aortic aneurysm growth predicted by uptake of ultrasmall superparamagnetic particles of iron oxide//Circulation. 2011. Vol. 4, N 3. Р. 274–281. doi: 10.1161/CIRCIMAGING.110.959866
- Trivedi R.A., U-King-Im J.M., Graves M.J., et al. In vivo detection of macrophages in human carotid atheroma: temporal dependence of ultrasmall superparamagnetic particles of iron oxide-enhanced MRI//Stroke. 2004. Vol. 35, N 7. Р. 1631–1635. doi: 10.1161/01.STR.0000131268.50418.b7
- McBride O.M., Berry C., Burns P., et al. MRI using ultrasmall superparamagnetic particles of iron oxide in patients under surveillance for abdominal aortic aneurysms to predict rupture or surgical repair: MRI for abdominal aortic aneurysms to predict rupture or surgery ― the MA 3 RS study//Open Heart. 2015. Vol. 2, N 1. Р. e000190. doi: 10.1136/openhrt-2014-000190
- Neubauer S. Cardiac magnetic resonance spectroscopy//Curr Cardiol Rep. 2003. Vol. 5, N 1. Р. 75–82. doi: 10.1007/s11886-003-0041-0
- Zajicek J., Pearlman J.D., Merickel M.B., et al. High-Resolution proton NMR spectra of human arterial plaque//Biochem Biophys Res Commun. 1987. Vol. 149, N 2. Р. 437–442. doi: 10.1016/0006-291X(87)90386-X
- Xin L., Lanz B., Lei H, Gruetter R. Assessment of metabolic fluxes in the mouse brain in vivo using 11 H-[ 13 C] NMR Spectroscopy at 14.1 Tesla//J Cereb Blood Flow Metab. 2015. Vol. 35, N 5. Р. 759–765. doi: 10.1038/jcbfm.2014.251
- Deelchand D.K., Moortele P.F., Adriany G., et al. In vivo 1H NMR spectroscopy of the human brain at 9.4T: initial results//J Magn Reson. 2010. Vol. 206, N 1. Р. 74–80. doi: 10.1016/j.jmr.2010.06.006
- Bentzon J.F., Otsuka F., Virmani R., Falk E. Mechanisms of plaque formation and rupture//Circ Res. 2014. Vol. 114, N 12. Р. 1852–1866. doi: 10.1161/CIRCRESAHA.114.302721
- Gholami S., Salavati A., Houshmand S., et al. Assessment of atherosclerosis in large vessel walls: a comprehensive review of FDG-PET/CT image acquisition protocols and methods for uptake quantification//J Nucl Cardiol. 2015. Vol. 22, N 3. Р. 468–479. doi: 10.1007/s12350-015-0069-8
- Folco E.J., Sheikine Y., Rocha V.Z., et al. Hypoxia but not inflammation augments glucose uptake in human macrophages: implications for imaging atherosclerosis with 18Fluorine-Labeled 2-deoxy-D-glucose positron emission tomography//J Am Coll Cardiol. 2011. Vol. 58, N 6. Р. 603–614. doi: 10.1016/j.jacc.2011.03.044
- Menezes L.J., Kotze C.W., Agu O., et al. Investigating vulnerable atheroma using combined 18F-FDG PET/CT angiography of carotid plaque with immunohistochemical validation//J Nucl Med. 2011. Vol. 52, N 11. Р. 1698–1703. doi: 10.2967/jnumed.111.093724
- Wykrzykowska J., Lehman S., Williams G., et al. Imaging of inflamed and vulnerable plaque in coronary arteries with 18F-FDG PET/CT in patients with suppression of myocardial uptake using a low-carbohydrate, high-fat preparation//J Nucl Med. 2009. Vol. 50, N 4. Р. 563–568. doi: 10.2967/jnumed.108.055616
- Græbe M., Pedersen S.F., Borgwardt L., et al. Molecular pathology in vulnerable carotid plaques: correlation with [18]-fluorodeoxyglucose positron emission tomography (FDG-PET)//Eur J Vasc Endovasc Surg. 2009. Vol. 37, N 6. Р. 714–721. doi: 10.1016/j.ejvs.2008.11.018
- Tarkin J.M., Dweck M.R., Evans N.R., et al. Imaging atherosclerosis//Circ Res. 2016. Vol. 118, N 4. Р. 750–769. doi: 10.1161/CIRCRESAHA.115.306247
- Malmberg C., Ripa R.S., Johnbeck C.B., et al. 64Cu-DOTATATE for noninvasive assessment of atherosclerosis in large arteries and its correlation with risk factors: head-to-head comparison with 68Ga-DOTATOC in 60 patients//J Nucl Med. 2015. Vol. 56, N 12. Р. 1895–1900. doi: 10.2967/jnumed.115.161216
- Dweck M.R., Aikawa E., Newby D.E., et al. Noninvasive molecular imaging of disease activity in atherosclerosis//Circ Res. 2016. Vol. 119, N 2. Р. 330–340. doi: 10.1161/CIRCRESAHA.116.307971
- Tarkin J.M., Joshi F.R., Evans N.R., et al. Detection of Atherosclerotic Inflammation by 68 Ga-DOTATATE PET Compared to [18 F] FDG PET Imaging//J Am Coll Cardiol. 2017. Vol. 69, N 14. Р. 1774–1791. doi: 10.1016/j.jacc.2017.01.060
- Gaemperli O., Shalhoub J., Owen D.R., et al. Imaging intraplaque inflammation in carotid atherosclerosis with 11C-PK11195 positron emission tomography/computed tomography//Eur Heart J. 2012. Vol. 33, N 15. Р. 1902–1910. doi: 10.1093/eurheartj/ehr367
- Weiberg D., Thackeray J.T., Daum G., et al. Clinical molecular imaging of chemokine receptor cxcr4 expression in atherosclerotic plaque using 88 Ga-Pentixafor PET: correlation with cardiovascular risk factors and calcified plaque burden//J Nucl Med. 2018. Vol. 59, N 2. Р. 266–272. doi: 10.2967/jnumed.117.196485
- Derlin T., Sedding D.G., Dutzmann J., et al. Imaging of chemokine receptor CXCR4 expression in culprit and nonculprit coronary atherosclerotic plaque using motion-corrected [68Ga]pentixafor PET/CT//Eur J Nucl Med Mol Imaging. 2018. Vol. 45, N 11. Р. 1934–1944. doi: 10.1007/s00259-018-4076-2
- Vesey A.T., Jenkins W.S., Irkle A., et al. 18F-Fluoride and 18F-fluorodeoxyglucose positron emission tomography after transient ischemic attack or minor ischemic stroke: Case-Control study//Circ Cardiovasc Imaging. 2017. Vol. 10, N 3. Р. e004976–e004976. doi: 10.1161/CIRCIMAGING.116.004976
- Joshi N.V., Vesey A.T., Williams M.C., et al. 18F-fluoride positron emission tomography for identification of ruptured and high-risk coronary atherosclerotic plaques: a prospective clinical trial//Lancet. 2014. Vol. 383, N 9918. Р. 705–713. doi: 10.1016/S0140-6736(13)61754-7
- Forsythe R.O., Dweck M.R., McBride O.M., et al. 18F-Sodium fluoride uptake in abdominal aortic aneurysms: the SoFIA3 study//J Am Coll Cardiol. 2018. Vol. 71, N 5. Р. 513–523. doi: 10.1016/j.jacc.2017.11.053
- Mateo J., Izquierdo-Garcia D., Badimon J.J., et al. Noninvasive assessment of hypoxia in rabbit advanced atherosclerosis using 18 f-fluoromisonidazole positron emission tomographic imaging//Circulation. 2014. Vol. 7, N 2. Р. 312–320. doi: 10.1161/CIRCIMAGING.113.001084
- Joshi F.R., Manavaki R., Fryer T.D., et al. Imaging of hypoxia and inflammation in carotid atherosclerosis with 18F-fluoromisonidazole and 18F-fluorodeoxyglucose positron emission tomography//Circulation. 2013. Vol. 128. Р. A14673.
- Winter P.M., Morawski A.M., Caruthers S.D., et al. Molecular imaging of angiogenesis in early-stage atherosclerosis with αvβ3-integrin–targeted nanoparticles//Circulation. 2003. Vol. 108, N 18. Р. 2270–2274. doi: 10.1161/01.CIR.0000093185.16083.95
- Haubner R., Kuhnast B., Mang C., et al. [18 F] Galacto-RGD: synthesis, radiolabeling, metabolic stability, and radiation dose estimates//Bioconjug Chem. 2004. Vol. 15, N 1. Р. 61–69. doi: 10.1021/bc034170n
- Laitinen I., Saraste A., Weidl E., et al. Evaluation of αVβ3 integrin-targeted positron emission tomography tracer 18F-Galacto-RGD for imaging of vascular inflammation in atherosclerotic MiceCLINICAL perspective//Circ Cardiovasc Imaging. 2009. Vol. 2, N 4. Р. 331–338. doi: 10.1161/CIRCIMAGING.108.846865
- Beer A.J., Pelisek J., Heider P., et al. Pet/Ct imaging of integrin αvβ3 expression in human carotid atherosclerosis//JACC Cardiovascular Imaging. 2014. Vol. 7, N 2. Р. 178–187. doi: 10.1016/j.jcmg.2013.12.003
- SCOT-HEART investigators. CT coronary angiography in patients with suspected angina due to coronary heart disease (SCOT-HEART): an open-label, parallel-group, multicentre trial//Lancet. 2015. Vol. 385, N 9985. Р. 2383–2391. doi: 10.1016/S0140-6736(15)60291-4
- Pugliese F., Mollet N.R., Runza G., et al. Diagnostic accuracy of non-invasive 64-slice CT coronary angiography in patients with stable angina pectoris//Eur Radiol. 2006. Vol. 16, N 3. Р. 575–582. doi: 10.1007/s00330-005-0041-0
- Williams M.C., Moss A.J., Dweck M., et al. Coronary artery plaque characteristics associated with adverse outcomes in the SCOT-HEART study//J Am Coll Cardiol. 2019. Vol. 73, N 3. Р. 291–301. doi: 10.1016/j.jacc.2018.10.066
- Greenland P., LaBree L., Azen S.P., et al. Coronary artery calcium score combined with Framingham score for risk prediction in asymptomatic individuals//JAMA. 2004. Vol. 291, N 2. Р. 210–215. doi: 10.1001/jama.291.2.210
- Polonsky T.S., McClelland R.L., Jorgensen N.W., et al. Coronary artery calcium score and risk classification for coronary heart disease prediction//JAMA. 2010. Vol. 303, N 16. Р. 1610–1616. doi: 10.1001/jama.2010.461
- Schuurman A.S., Vroegindewey M., Kardys I., et al. Near-Infrared spectroscopy-derived lipid core burden index predicts adverse cardiovascular outcome in patients with coronary artery disease during long-term follow-up//Eur Heart J. 2018. Vol. 39, N 4. Р. 295–302. doi: 10.1093/eurheartj/ehx247
- Dweck M.R., Chow M.W., Joshi N.V., et al. Coronary arterial 18F-Sodium fluoride uptake: a novel marker of plaque biology//J Am Coll Cardiol. 2012. Vol. 59, N 17. Р. 1539–1548. doi: 10.1016/j.jacc.2011.12.037
- Dalm V.A., van Hagen P.M., van Koetsveld P.M., et al. Expression of somatostatin, cortistatin, and somatostatin receptors in human monocytes, macrophages, and dendritic cells//Am J Physiol Endocrinol Metab. 2003. Vol. 285, N 2. Р. E344–E353. doi: 10.1152/ajpendo.00048.2003
- Gasecki A.P., Eliasziw M., Ferguson G.G., et al. Long-Term prognosis and effect of endarterectomy in patients with symptomatic severe carotid stenosis and contralateral carotid stenosis or occlusion: results from NASCET. North American Symptomatic Carotid Endarterectomy Trial (NASCET) Group//J Neurosurg. 1995. Vol. 83, N 5. Р. 778–782. doi: 10.3171/jns.1995.83.5.0778
- Randomised trial of endarterectomy for recently symptomatic carotid stenosis: final results of the MRC European carotid surgery trial (ECST)//Lancet. 1998. Vol. 351, N 9113. Р. 1379–1387. doi: 10.1016/S0140-6736(97)09292-1
- Rothwell P.M., Eliasziw M., Gutnikov S.A., et al. Endarterectomy for symptomatic carotid stenosis in relation to clinical subgroups and timing of surgery//Lancet. 2004. Vol. 363, N 9413. Р. 915–924. doi: 10.1016/S0140-6736(04)15785-1
- Oates C.P., Naylor A.R., Hartshorne T., et al. Joint recommendations for reporting carotid ultrasound investigations in the United Kingdom//Eur J Vasc Endovasc Surg. 2009. Vol. 37, N 3. Р. 251–261. doi: 10.1016/j.ejvs.2008.10.015
- Brott T.G., Halperin J.L., Abbara S., et al. ASA/ACCF/AHA/AANN/AANS/ACR/ASNR/CNS/SAIP/SCAI/SIR/SNIS/SVM/SVS guideline on the management of patients with extracranial carotid and vertebral artery disease: a report of the American College of cardiology Foundation/American heart association Task force on practice guidelines, and the American stroke association, American association of neuroscience nurses, American association of neurological Surgeons, American College of radiology, American Society of Neuroradiology, Congress of neurological Surgeons, society of atherosclerosis imaging and prevention, Society for cardiovascular angiography and interventions, society of interventional radiology, society of NeuroInterventional surgery, Society for vascular medicine, and Society for vascular surgery developed in collaboration with the American Academy of Neurology and society of cardiovascular computed tomography//J Am Coll Cardiol. 2011. Vol. 57, N 8. Р. e16–94. doi: 10.1016/j.jacc.2010.11.006
- Spence J.D., Naylor A.R., Endarterectomy N.A. Endarterectomy, stenting, or neither for asymptomatic carotid-artery stenosis//N Engl J Med. 2016. Vol. 374, N 11. Р. 1087–1088. doi: 10.1056/NEJMe1600123
- Forsythe R.O., Newby D.E., Robson J.M. Monitoring the biological activity of abdominal aortic aneurysms Beyond Ultrasound//Heart. 2016. Vol. 102, N 11. Р. 817–824. doi: 10.1136/heartjnl-2015-308779
- Duddalwar V.A. Multislice CT angiography: a practical guide to CT angiography in vascular imaging and intervention//Br J Radiol. 2004. Vol. 77, Suppl 1. Р. S27–S38. doi: 10.1259/bjr/25652856
- Castrucci M., Mellone R., Vanzulli A., et al. Mural thrombi in abdominal aortic aneurysms: MR imaging characterization--useful before endovascular treatment?//Radiology. 1995. Vol. 197, N 1. Р. 135–139. doi: 10.1148/radiology.197.1.7568811
- Nguyen V.L., Leiner T., Hellenthal F.A., et al. Abdominal aortic aneurysms with high thrombus signal intensity on magnetic resonance imaging are associated with high growth rate//Eur J Vasc Endovasc Surg. 2014. Vol. 48, N 6. Р. 676–684. doi: 10.1016/j.ejvs.2014.04.025
- Derlin T., Tóth Z., Papp L., et al. Correlation of inflammation assessed by 18F-FDG PET, active mineral deposition assessed by 18F-fluoride PET, and vascular calcification in atherosclerotic plaque: a dual-tracer PET/CT study//J Nucl Med. 2011. Vol. 52, N 7. Р. 1020–1027. doi: 10.2967/jnumed.111.087452
- Janssen T., Bannas P., Herrmann J., et al. Association of linear 18F-sodium fluoride accumulation in femoral arteries as a measure of diffuse calcification with cardiovascular risk factors: a PET/CT study//J Nucl Cardiol. 2013. Vol. 20, N 4. Р. 569–577. doi: 10.1007/s12350-013-9680-8
- Chhatriwalla A.K., Nicholls S.J., Nissen S.E. The asteroid trial: coronary plaque regression with high-dose statin therapy//Future Cardiol. 2006. Vol. 2, N 6. Р. 651–654. doi: 10.2217/14796678.2.6.651
- Nissen S.E., Tuzcu E.M., Schoenhagen P., et al. Effect of intensive compared with moderate lipid-lowering therapy on progression of coronary atherosclerosis//JAMA. 2004. Vol. 291, N 9. Р. 1071–1080. doi: 10.1001/jama.291.9.1071
- Okazaki S., Yokoyama T., Miyauchi K., et al. Early statin treatment in patients with acute coronary syndrome: demonstration of the beneficial effect on atherosclerotic lesions by serial volumetric intravascular ultrasound analysis during half a year after coronary event: the establish study//Circulation. 2004. Vol. 110, N 9. Р. 1061–1068. doi: 10.1161/01.CIR.0000140261.58966.A4
- Underhill H.R., Yuan C., Zhao X.Q., et al. Effect of rosuvastatin therapy on carotid plaque morphology and composition in moderately hypercholesterolemic patients: a high-resolution magnetic resonance imaging trial//Am Heart J. 2008. Vol. 155, N 3. Р. 584.e1–584.e8. doi: 10.1016/j.ahj.2007.11.018
- Zhao X.Q., Dong L., Hatsukami T., et al. Mr imaging of carotid plaque composition during lipid-lowering therapy: a prospective assessment of effect and time course//JACC Cardiovasc Imaging. 2011. Vol. 4, N 9. Р. 977–986. doi: 10.1016/j.jcmg.2011.06.013
- Tang T.Y., Howarth S.P., Miller S.R., et al. The atheroma (atorvastatin therapy: effects on reduction of macrophage activity) study: evaluation using ultrasmall superparamagnetic iron oxide-enhanced magnetic resonance imaging in carotid disease//J Am Coll Cardiol. 2009. Vol. 53, N 22. Р. 2039–2050. doi: 10.1016/j.jacc.2009.03.018
- Schoenhagen P., Tuzcu E.M., Apperson-Hansen C., et al. Determinants of arterial wall remodeling during lipid-lowering therapy: serial intravascular ultrasound observations from the reversal of atherosclerosis with aggressive lipid lowering therapy (reversal) trial//Circulation. 2006. Vol. 113, N 24. Р. 2826–2834. doi: 10.1161/CIRCULATIONAHA.105.585703
- Zimarino M., Angeramo F., Prasad A., et al. Reduction of atherothrombotic burden before stent deployment in non-ST elevation acute coronary syndromes: reduction of myocardial necrosis achieved with nose-dive manual thrombus aspiration (remnant) trial. A volumetric intravascular ultrasound study//Cathet Cardiovasc Intervent. 2016. Vol. 88, N 5. Р. 716–725. doi: 10.1002/ccd.26301
- Fayad Z.A., Mani V., Woodward M., et al. Safety and efficacy of dalcetrapib on atherosclerotic disease using novel non-invasive multimodality imaging (dal-PLAQUE): a randomised clinical trial//Lancet. 2011. Vol. 378, N 9802. Р. 1547–1559. doi: 10.1016/S0140-6736(11)61383-4.
Дополнительные файлы