Рекомендации CARE для описания случаев: разъяснения и уточнения
- Авторы: Barber M.S.1, Aronson J.K.2, von Schoen-Angerer T.3,4, Riley D.S.1,5, Tugwell P.6, Kiene H.7, Helfand M.8, Altman D.G.2, Sox H.2, Werthmann P.G.7, Moher D.9, Rison R.A.10, Shamseer L.11, Koch C.A.12, Sun G.H.13, Hanaway P.14,15, Sudak N.L.16, Carpenter J.E.17, Gagnier J.J.18,19
-
Учреждения:
- Integrative Medicine Institute
- University of Oxford
- Centre medical de La Chapelle, Chemin de Compostelle
- ACIM Institute, Filderklinik, Im Haberschlai
- Helfgott Research Institute
- University of Ottawa,451 Smyth Rd, Ottawa, ON
- Institute for Applied Epistemology and Medical Methodology at the University of Witten-Herdecke
- 3710 SW US Veterans Hospital
- Ottawa Methods Centre, Ottawa Hospital Research Institute
- PIH Health Hospital-Whittier, Neurology Consultants Medicine Group, University of Southern California Keck School of Medicine
- Ottawa Hospital Research Institute
- University of Mississippi Medical Center
- Rancho Los Amigos National Rehabilitation Center
- Geisel School of Medicine at Dartmouth
- Cener for Functional Medicine, Cleveland Clinic
- PharmaLex Harrlachweg
- 24 Frank Lloyd Wright Drive, Lobby A, Ann Arbor, MI 48106
- School of Public Health 1415 Washington Heights
- 24 Frank Lloyd Wright Drive
- Выпуск: Том 3, № 1 (2022)
- Страницы: 16-42
- Раздел: Научные обзоры
- URL: https://journals.rcsi.science/DD/article/view/105291
- DOI: https://doi.org/10.17816/DD105291
- ID: 105291
Цитировать
Полный текст
Аннотация
Обоснование. Доступное и понятное описание клинических случаев помогает выявлять самые ранние данные о возможной пользе, неблагоприятном влиянии и расходовании ресурсов; предоставляет информацию для клинической научной работы и разработки рекомендаций для клинической практики; интегрируется в медицинское образование. Авторы с большей вероятностью подготовят высококачественные описания случаев, если они будут следовать определённым правилам написания таких публикаций. В 2011–2012 гг. группа клиницистов, учёных и редакторов журналов разработала рекомендации для точного представления информации при описании случаев, итогом которых стало Положение и проверочный перечень CARE (CAse Report ― описание случаев), представленных в 2013 г. на Международном конгрессе по экспертному рецензированию и биомедицинским публикациям, поддержанных многочисленными медицинскими журналами и переведённых на девять языков.
Цель этого разъясняющего и уточняющего документа ― более широко внедрить и распространить использование проверочного перечня CARE при подготовке и опубликовании описаний случаев.
Схема (дизайн) статьи и контекст. К каждому пункту проверочного перечня CARE даны пояснения и прилагаются примеры из публикаций. Объяснения и примеры в данном документе помогают авторам подготовить высококачественное описание случая, а редакторам, экспертам-рецензентам и читателям ― критически проанализировать его.
Результаты и выводы. Данная статья вместе с Положением и проверочным перечнем CARE от 2013 г., с которым можно ознакомиться на веб-сайте CARE (www.care-statement.org) и веб-сайте группы EQUATOR (www.equator-network.org), служит способом улучшить полноту и прозрачность описания случаев.
Источник. Данная статья является переводом оригинальной публикации «CARE guidelines for case reports: explanation and elaboration document» в Journal of Clinical Epidemiology (doi: 10.1016/j.jclinepi.2017.04.026), созданным с разрешения правообладателя (Elsevier Inc.), под руководством научного редактора и переводчика профессора, д.м.н. Е.Г. Старостиной (Москва, Россия).
Ключевые слова
Полный текст
Открыть статью на сайте журналаОб авторах
Melissa S. Barber
Integrative Medicine Institute
Автор, ответственный за переписку.
Email: dsriley@care-statement.org
США, 2437A NW Overton Street, OR 97210, Портленд
Jeffrey K. Aronson
University of Oxford
Email: dsriley@care-statement.org
ORCID iD: 0000-0003-1139-655X
Nuffield Department of Primary Care Health Sciences, Radcliffe Observatory Quarter
Великобритания, Woodstock Road, OX2 6GG, ОксфордTido von Schoen-Angerer
Centre medical de La Chapelle, Chemin de Compostelle; ACIM Institute, Filderklinik, Im Haberschlai
Email: dsriley@care-statement.org
ORCID iD: 0000-0003-3601-7480
Department of Pediatrics
Швейцария, 7,1212 Grand-Lancy, Женева; 7,70794 Filderstadt-Bonlanden, ФильдерштадтDavid S. Riley
Integrative Medicine Institute; Helfgott Research Institute
Email: dsriley@care-statement.org
ORCID iD: 0000-0001-6903-6624
США, 2437A NW Overton Street, Портленд, OR 97210; 2220 SW 1st Ave, Портленд, OR 97201
Peter Tugwell
University of Ottawa,451 Smyth Rd, Ottawa, ON
Email: dsriley@care-statement.org
ORCID iD: 0000-0001-5062-0556
Department of Medicine
Канада, K1H 8M5, ОттаваHelmut Kiene
Institute for Applied Epistemology and Medical Methodology at the University of Witten-Herdecke
Email: dsriley@care-statement.org
Senior Research Scientist
Германия, Zechenweg 6,79111, Фрайбург-им-БрайсгауM. Helfand
3710 SW US Veterans Hospital
Email: dsriley@care-statement.org
ORCID iD: 0000-0003-4846-9900
Departmenty of Medical Informatics and C linical Epidemiology
США, OR 97239, ПортлендDouglas G. Altman
University of Oxford
Email: dsriley@care-statement.org
ORCID iD: 0000-0002-7183-4083
Center for Statistics ― Botnar Research Centre
Великобритания, Windmill Road, OX3 7LD, ОксфордH. Sox
University of Oxford
Email: dsriley@care-statement.org
Center for Statistics ― Botnar Research Centre, Windmill Road
Великобритания, OX3 7LD, ОксфордPaul G. Werthmann
Institute for Applied Epistemology and Medical Methodology at the University of Witten-Herdecke
Email: dsriley@care-statement.org
ORCID iD: 0000-0002-1808-7787
Senior Research Scientist
Германия, Zechenweg 6,79111, Фрайбург-им-БрайсгауDavid Moher
Ottawa Methods Centre, Ottawa Hospital Research Institute
Email: dsriley@care-statement.org
ORCID iD: 0000-0003-2434-4206
Senior Scientist
Канада, 501 Smythe Road, ON, K1H 8L6, ОттаваRichard A. Rison
PIH Health Hospital-Whittier, Neurology Consultants Medicine Group, University of Southern California Keck School of Medicine
Email: dsriley@care-statement.org
ORCID iD: 0000-0002-6088-8246
США, 12291 Washington Blvd # 303, Whittier, CA 90606, Лос-Анджелес
Larissa Shamseer
Ottawa Hospital Research Institute
Email: dsriley@care-statement.org
ORCID iD: 0000-0003-3690-3378
Senior Scientist, Ottawa Methods Centre
Канада, 501 Smythe Road, Ottawa, ON, Canada K1H 8L6, ОттаваChristian A. Koch
University of Mississippi Medical Center
Email: dsriley@care-statement.org
ORCID iD: 0000-0003-0678-1242
Professor of Medicine, Director ― Endocrinology
США, 2500 N. State Street, MS 39216, ДжексонGordon H. Sun
Rancho Los Amigos National Rehabilitation Center
Email: dsriley@care-statement.org
Medical Director of Inpatient Services
США, 7601 Imperial Highway, Downey, CA 90242, ДауниPatrick Hanaway
Geisel School of Medicine at Dartmouth; Cener for Functional Medicine, Cleveland Clinic
Email: dsriley@care-statement.org
ORCID iD: 0000-0002-7788-783X
Professor Medicine and of The Dartmouth Institute
США, 31 Faraway Lane, NH 03784-4401, Западный Ливан; H-18,9500 Euclid Avenue, OH,44195, КливлендNancy L. Sudak
PharmaLex Harrlachweg
Email: dsriley@care-statement.org
ORCID iD: 0000-0002-6515-2780
Director of Scientific Affairs
Германия, 6, 68163, МангеймJames E. Carpenter
24 Frank Lloyd Wright Drive, Lobby A, Ann Arbor, MI 48106
Email: dsriley@care-statement.org
ORCID iD: 0000-0003-3890-6206
Department Chair, Orthopaedic Surgery
США, Анн-АрборJoel J. Gagnier
School of Public Health 1415 Washington Heights; 24 Frank Lloyd Wright Drive
Email: dsriley@care-statement.org
ORCID iD: 0000-0002-3162-3935
Department of Orthopaedic Surgery, Department of Epidemiology
США, MI 48109-2029, Анн-Арбор; Lobby A, Анн-Арбор, MI 48106Список литературы
- Vandenbroucke J.P. In defense of case reports and case series // Ann Intern Med. 2001. Vol. 134, N 4. Р. 330–334. doi: 10.7326/0003-4819-134-4-200102200-00017
- Aronson J.K. Anecdotes as evidence // BMJ 2003. Vol. 326, N 2. Р. 1346. doi: 10.1111/j.1365-2753.2005.00527.x
- Sun G.H., Aliu O., Hayward R.A. Open-access electronic case report journals: the rationale for case report guidelines // J Clin Epidemiol. 2013. Vol. 66, N 10. Р. 1065–1070. doi: 10.1016/j.jclinepi.2013.04.001
- Akers K.G. New journal for publishing medical case reports // J Med Libr Assoc. 2016. Vol. 104, N 2. Р. 146–149. doi: 10.3163/1536-5050.104.2.010
- McBride W.G. Thalidomide and congenital abnormalities // Lancet 1961. Vol. 278, N 7216. Р. 1358. doi: 10.1016/S0140-6736(61)90927-8
- CDC. Pneumocystis pneumonia e Los Angeles // Morbidity Mortality Weekly Rep. 1981. Vol. 30, N 21. Р. 250–252.
- Rison R., Helfgott M. Acute paradoxical embolic cerebral ischemia secondary to possible May-Thurner syndrome and an atrial septal defect: a case report // J Med Case Rep. 2013. Vol. 7. Р. 172. doi: 10.1186/1752-1947-7-172
- Driggers R.W., Ho C.Y., Korhonen E.M., et al. Zika virus infection with prolonged maternal viremia and fetal brain abnormalities // N Engl J Med. 2016. Vol. 374, N 22. Р. 2142–2151. doi: 10.1056/NEJMoa1601824
- Kaszkin-Bettag M., Hildebrandt W. Case report on cancer therapies: the urgent need to improve the reporting quality // Glob Adv Health Med. 2012. Vol. 1, N 2. Р. 8–10. doi: 10.7453/gahmj.2012.1.2.002
- Richason T.P., Paulson S.M., Lowenstein S.R., Heard K.J. Case reports describing treatments in the emergency medicine literature: missing and misleading information // BMC Emerg Med. 2009. Vol. 9. Р. 10. doi: 10.1186/1471-227X-9-10
- Moher D., Schulz K.F., Simera I., Altman D.G. Guidance for developers of health research reporting guidelines // Plos Med. 2010. Vol. 7, N 2. Р. e1000217. doi: 10.1371/journal.pmed.1000217
- Gagnier J.J., Kienle G., Altman D.G., et al.; The CARE Group. The CARE guidelines: consensus-based clinical case report guideline development // Glob Adv Health Med. 2013. Vol. 2, N 5. Р. 38–43. doi: 10.7453/gahmj.2013.008
- Galipeau J., Barbour V., Baskin P., et al. A scoping review of competencies for scientific editors of biomedical journals // BMC Med. 2016. Vol. 14. Р. 16. doi: 10.1186/s12916-016-0561-2
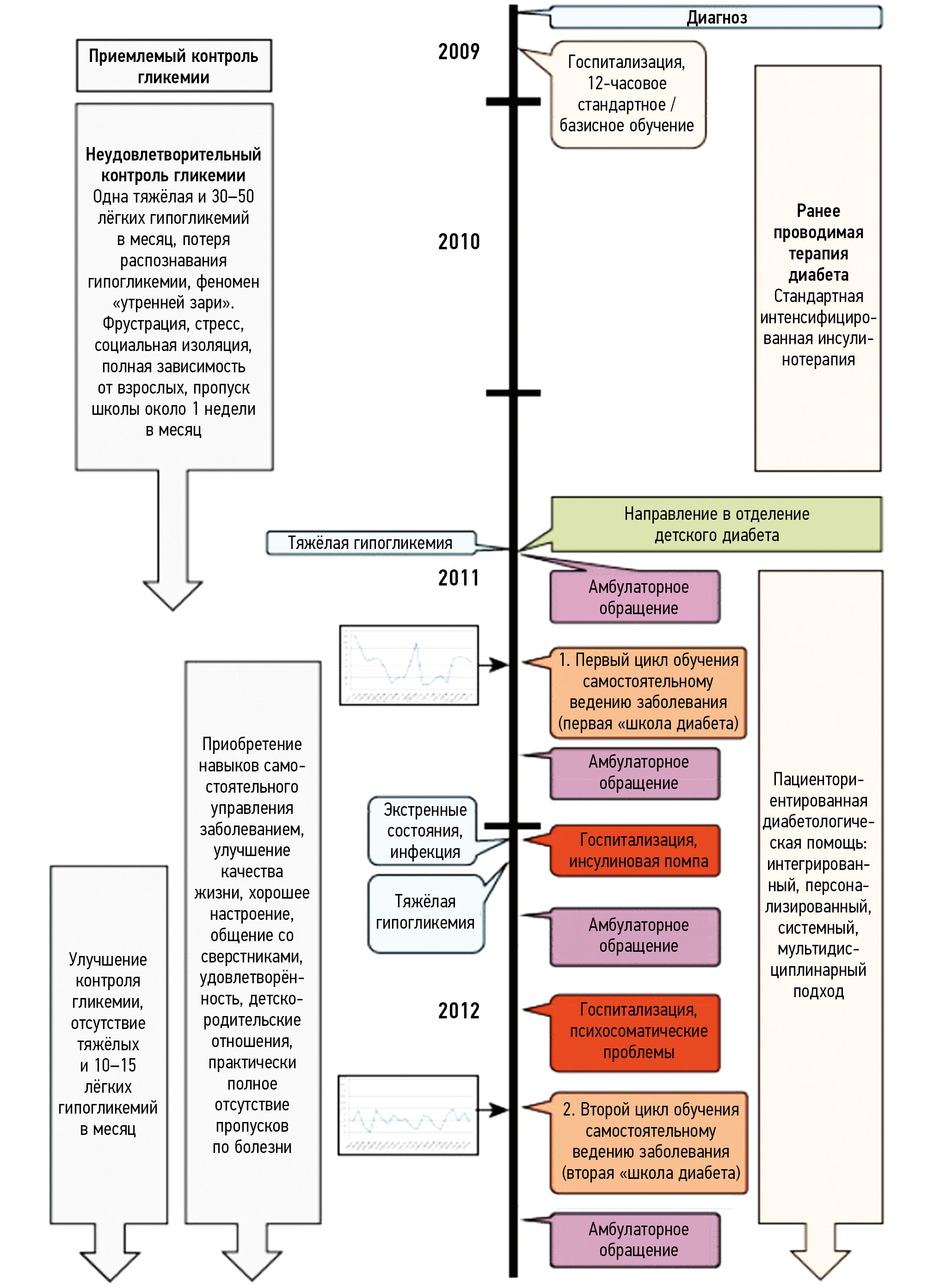
- Kienle G.S., Meusers M., Quecke B., Hilgard D. Patient-centered Diabetes care in children: an integrated, Individualized, Systemsoriented, and multi-Disciplinary Approach // Glob Adv Health Med. 2013. Vol. 2, N 2. Р. 12–19. doi: 10.7453/gahmj.2013.005
- Jenicek M. Clinical case reporting in evidence-based medicine. 2nd ed. London/New York: Arnold and Oxford University Press,2001.231 р.
- Wei J., Chang C.Y., Chuang Y.C., et al. Successful heart transplantation after 13 hours of donor heart ischemia with the Use of HTK Solution: a case report // Transplant Proc 2005. Vol. 37, N 5. Р. 2253–2254. doi: 10.1016/j.transproceed.2005.03.055
- Gish D.S., Baer J.A., Crabtree G.S., et al. Impending aortic aneurysm rupture ― a case report and review of the warning signs // J Community Hosp Intern Med Perspect. 2016. Vol. 6, N 5. Р. 32217. doi: 10.3402/jchimp.v6.32217
- Cooper D. How to write an original research paper (and get it published) // J Med Lib Assoc. 2015. Vol. 103, N 2. Р. 67–68. doi: 10.3163/1536-5050.103.2.001
- Jha K.N. How to write articles that get published // J Clin Diagn Res. 2014. Vol. 8, N 9. Р. XG01–XG03. doi: 10.7860/JCDR/2014/8107.4855
- Cohen H. How to write a patient case report // Am J HealthSyst Pharm. 2006. Vol. 63, N 19. Р. 1888–1892. doi: 10.2146/ajhp060182
- Hopewell S., Clarke M., Moher D., et al. CONSORT for reporting randomized controlled trials in journal and Conference abstracts: explanation and elaboration // Plos Med. 2008. Vol. 5, N 1. Р. e20. doi: 10.1371/journal.pmed.0050020
- Davidson A., Delbridge E. How to write a research paper // Paediatrics Child Health. 2012. Vol. 22, N 2. Р. 61–65. doi: 10.1016/j.paed.2011.05.009
- Vandenbroucke J. Case reports in an evidence-based world // JR Soc Med. 1999. Vol. 92, N 4. Р. 159–162. doi: 10.1177/014107689909200401
- Barre-Sinoussi F., Chermann J.C., Rey F., et al. Isolation of a t-lymphotropic retrovirus from a patient at risk for acquired immune deficiency syndrome (aids) // Science. 1983. Vol. 220, N 4599. Р. 868–871. doi: 10.1126/science.6189183
- Labris G., Katsanos A., Karapetsa M., et al. Association between isotretinoin use and central retinal vein occlusion in an adolescent with minor predisposition for thrombotic incidents: a case report // J Med Case Rep. 2009. Vol. 3. Р. 58. doi: 10.1186/1752-1947-3-58
- Green B.N., Johnson C.D. How to write a case report for publication // J Chiropr Med. 2006. Vol. 5, N 2. Р. 72–82. doi: 10.1016/S0899-3467(07)60137-2
- Rison R.A. A guide to writing case reports for the journal of medical case reports and BioMed central research notes // J Med Case Rep. 2013. Vol. 7. Р. 239. doi: 10.1186/1752-1947-7-239
- Turner L., Shamseer L., Altman D.G., et al. Does use of the CONSORT Statement impact the completeness of reporting of randomized controlled trials published in medical journals? A Cochrane review // Syst Rev. 2012. Vol. 1. Р. 60. doi: 10.1186/2046-4053-1-60
- Chang H.C., Burbridge H., Wong C. Extensive deep vein thrombosis following prolonged gaming (‘gamer’s thrombosis’): a case report // J Med Case Rep. 2013. Vol. 7. Р. 235. doi: 10.1186/1752-1947-7-235
- Gottlieb M., Schroff R., Schanker H.M., et al. Pneumocystis carinii pneumonia and mucosal candidiasis in previously health homosexual men; evidence of a new acquired cellular immunodeficiency // N Engl J Med. 1981. Vol. 305, N 24. Р. 1425–1431. doi: 10.1056/NEJM198112103052401
- Lang T.A., Talerico C., Siontis G.C. Documenting clinical and laboratory images in publications: the CLIP principles // Chest. 2012. Vol. 141, N 16. Р. 1626–1632. doi: 10.1378/chest.11-1800
- Dahlbäck B., Carlsson M., Svensson P. Familial thrombophilia due to a previously unrecognized mechanism characterized by poor anticoagulant response to activated protein C: Prediction of a cofactor to activated protein C // Proc Natl Acad Sci. 1993. Vol. 90, N 3. Р. 1004. doi: 10.1073/pnas.90.3.1004
- Namas R., Marquardt A. Case report and literature review: Quinacrine-induced Cholestatic Hepatitis in undifferentiated connective tissue disease // J Rheumatol. 2015. Vol. 42, N 7. Р. 1354–1355. doi: 10.3899/jrheum.150050
- Rajakumar D., Rosenberg A.M. Mycobacterium tuberculosis monoarthritis in a child // Pediatr Rheumatol Online J. 2008. Vol. 6. Р. 15. doi: 10.1186/1546-0096-6-15
- Sevinc M., Basturk T., Sahutoglu T., et al. Plasma resistant atypical hemolytic uremic syndrome associated with a CFH mutation treated with eculizumab: a case report // J Med Case Rep. 2015. Vol. 9. Р. 92. doi: 10.1186/s13256-015-0575-y
- Cella D., Stone A.A. Health-related quality of life measurement in oncology: Advances and opportunities // Am Psychol. 2015. Vol. 70, N 2. Р. 175–185. doi: 10.1037/a0037821
- Hill A.B. The Environment and Disease: Association or Causation? // Proc R Soc Med. 1965. Vol. 58, N 5. Р. 295–300.
- Rison R.A. Branch facial nerve trauma after superficial temporal artery biopsy: a case report // J Med Case Rep. 2011. Vol. 5. Р. 34. doi: 10.1186/1752-1947-5-34
- Mora J.R., Rison R.A., Beudoun S.R. Chiari malformation type I with cervicothoracic syringomyelia masquerading as bibrachial amyotrophy: a case report // J Med Case Rep. 2015. Vol. 9. Р. 11. doi: 10.1186/1752-1947-9-11
- Gatselis N.K., Skendros P., Ritis K., Dalekos G.N. Severe liver involvement in two patients with long-term history of fever: remember familial Mediterranean fever // BMJ Case Rep. 2016. Vol. 2016. Р. bcr2016216941. doi: 10.1136/bcr-2016-216941
- Liu Y.J., Du P., Rao J. Procalcitonin as a diagnostic and prognostic marker for sepsis caused by intestinal infection: a case report // Eur Rev Med Pharmacol Sci. 2013. Vol. 17, N 10. Р. 1311–1313.
- Hoffmann T.C., Glasziou P.P., Boutron I., et al. Better reporting of interventions: template for intervention description and replication (TIDieR) Checklist and guide // BMJ. 2014. Vol. 7, N 3. Р. g1687. doi: 10.1055/s-0037-1600948
- Morino T., Ogata T., Horiuchi H., et al. Eight years of follow-up after laminectomy of calcium pyrophosphate crystal deposition in the cervical yellow ligament of patient with CoffineLowry syndrome: a case report // Medicine (Baltimore). 2016. Vol. 95, N 31. Р. e4468. doi: 10.1097/MD.0000000000004468
- Thomson N., Ulrickson M. Maintenance eculizumab dose adjustment in the treatment of atypical hemolytic uremic syndrome: a case report and review of the literature // Clin Case Rep. 2016. Vol. 4, N 8. Р. 773–776. doi: 10.1002/ccr3.628
- Tucker M., Kekulawala S., Kent M., et al. Polysubstance- induced relapse of schizoaffective disorder refractory to high-dose antipsychotic medications: a case report // J Med Case Rep. 2016. Vol. 10, N 1. Р. 242. doi: 10.1186/s13256-016-1031-3
- Gupta S., Szerszen A., Nakhl F., et al. Severe refractory autoimmune hemolytic anemia with both warm and cold autoantibodies that responded completely to a single cycle of rituximab: a case report // J Med Case Rep. 2011. Vol. 5. Р. 156. doi: 10.1186/1752-1947-5-156
- Oberg E., Rempe M., Bradley R. Self-directed mindfulness training and improvement in blood pressure, migraine frequency, and quality of life // Glob Adv Health Med. 2013. Vol. 2, N 2. Р. 20–25. doi: 10.7453/gahmj.2013.006
- Waqas M., Hadi Y.B., Sheikh S., Shamin S.M. Acute cerebellitis successful managed with temporary cerebrospinal fluid diversion using a long tunnel external ventricular drain EVD: a long-term radiological follow-up of two cases // BMJ Case Rep. 2016. Vol. 2016. Р. bcr2016215631. doi: 10.1136/bcr-2016-215631
- Tamanna S., Ullah M.I., Pope C.R., et al. Quetiapine- induced sleep-related eating disorder like behavior: a case series // J Med Case Rep. 2012. Vol. 6. Р. 380. doi: 10.1186/1752-1947-6-380
- Aronson J.K., Grahame-Smith D.G. Altered distribution of digoxin in renal failureda case of digoxin toxicity? // Br J Clin Pharmac. 1976. Vol. 3, N 6. Р. 1045–1051. doi: 10.1111/j.1365-2125.1976.tb00356.x
- Hauben M., Aronson J.K. Gold standards in pharmaco-vigilance: the use of definitive anecdotal reports of adverse drug reactions as pure gold and high-grade ore // Drug Saf. 2007. Vol. 30. Р. 645–655. doi: 10.2165/00002018-200730080-00001
- Wicomb W.N., Cooper D.K., Novitzky D., Barnard C.N. Cardiac transplantation following storage of the donor heart by a portable hypothermic perfusion system // Ann Thorac Surg. 1984. Vol. 37, N 8. Р. 243. doi: 10.1016/s0003-4975(10)60333-5
- Morgan J.A., John R., Weinberg A.D., et al. Prolonged donor ischemic time does not adversely affect long-term survival in adult patients undergoing cardiac transplantation // J Thorac Cardiovasc Surg. 2003. Vol. 126, N 5. Р. 1624–1633. doi: 10.1016/s0022-5223(03)01026-2
- Briganti E.M., Bergin P.J., Rosenfeldt F.L., et al. Successful long-term outcome with prolonged ischemic time cardiac allografts // J Heart Lung Transpl. 1995. Vol. 14, N 5. Р. 840–845.
- Del Rizzo D.F., Menkis A.H., Pflugfelder P.W., et al. The role of donor age and ischemic time on survival following orthotopic heart transplantation // J Heart Lung Transpl. 1999. Vol. 18, N 4. Р. 310–319. doi: 10.1016/s1053-2498(98)00059-x
- Canter C., Naftel D., Caldwell R., et al. Survival and risk factors for death after cardiac transplantation in infants: a multi-institutional study // Circ J. 1997. Vol. 96, N 1. Р. 227. doi: 10.1161/01.cir.96.1.227
- Keogh M.J., Findlay J.M., Leach S., Bowen J. Statin-associated weakness in myasthenia gravis: a case report // J Med Case Rep. 2010. Vol. 4. Р. 61. doi: 10.1186/1752-1947-4-61
- Dickman H., Fletke K., Redfern R.E. Prolonged unassisted survival in an infant with anencephaly // BMJ Case Rep. 2016. Vol. 2016. Р. bcr2016215986. doi: 10.1136/bcr-2016-215986
- Lundberg G.D. Sciatica: a love story. Medscape; 2016.
- Kogan M., Castillo C.C., Barber M.S. Chronic Rhinosinusitis and Irritable Bowel syndrome: a case report // Integr Med (encinitas) J. 2016. Vol. 15, N 3. Р. 44–48.
- BMJ Case Reports. Режим доступа: http://casereports.bmj.com/site/about/guidelines.xhtml#patientconsent. Дата обращения: 22.10.2016.
- Sackett D.L., Rosenberg W.M., Gray J.A., et al. Evidence basedmedicine: what it is andwhat it isn’t // BMJ. 1996. Vol. 312, N 7023. Р. 71. doi: 10.1136/bmj.312.7023.71
- Califf R.M., Robb M.A., Bindman A.B., et al. Transforming generation to support health and health care decisions // N Engl J Med. 2016. Vol. 375, N 24. Р. 2395–2440. doi: 10.1056/NEJMsb1610128
- Wacholder S., Chanock S., Garcia-Closas M.E., et al. Assessing the probability that a positive report is false: an Approach for molecular Epidemiology studies // J Natl Cancer Inst. 2004. Vol. 96, N 6. Р. 434–442. doi: 10.1093/jnci/djh075
- Ioannidis J.P. Why most clinical research is not useful // Plos Med. 2016. Vol. 13, N 6. Р. e1002049. doi: 10.1371/journal.pmed.1002049
- Buonfrate D., Requena-Mendez A., Angheben A., et al. Severe strongyloidiasis: a systematic review of case reports // BMC Infect Dis. 2013. Vol. 13. Р. 78. doi: 10.1186/1471-2334-13-78
- IOM (Institute of Medicine). Clinical Practice Guidelines We Can Trust. Washington, DC: The National Academies Press,2011.
- Barrows H.S. A taxonomy of problem-based learning methods // Med Education. 1986. Vol. 20, N 6. Р. 481–486. doi: 10.1111/j.1365-2923.1986.tb01386.x
- Irby D.M. Three Exemplary model of case-based teaching // Acad Med. 1994. Vol. 69, N 12. Р. 947–953. doi: 10.1097/00001888-199412000-00003
- Kiene H., Hamre H., Kienle G.S. In support of clinical case reports: a system of causality assessment // Glob Adv Health Med. 2013. Vol. 2, N 2. Р. 64–75. doi: 10.7453/gahmj.2012.061
- Hay J.E., Wiesner R.H., Shorter R.G., et al. Primary sclerosing cholangitis and celiac disease. A novel association // Ann Intern Med. 1988. Vol. 109, N 9. Р. 713–717. doi: 10.7326/0003-4819-109-9-713
- Shamseer L., Sampson M., Bukutu C., et al. Cent group. CONSORT extension for reporting N-of-1 trials (CENT) 2015: explanation and elaboration // J Clin Epidemiol. 2016. Vol. 76. Р. 18–46. doi: 10.1016/j.jclinepi.2015.05.018
- Badalov N., Baradarian R., Iswara K., et al. Drug-induced acute pancreatitis: an evidence-based review // Clin Gastroenterol Hepatol. 2007. Vol. 5, N 6. Р. 548–561. doi: 10.1016/j.cgh.2006.11.023
- Patsopoulos N.A., Analatos A.A., Ioannidis J.P. Relative citation impact of various study design in the health sciences // JAMA. 2005. Vol. 293, N 19. Р. 2362–2366
Дополнительные файлы