Роль магнитно-резонансной томографии в выявлении злокачественных лёгочных узлов: систематический обзор и метаанализ
- Авторы: Васильев Ю.А.1,2, Панина О.Ю.1,2,3, Грик Е.А.3, Ахмад Е.С.1, Васильева Ю.Н.3
-
Учреждения:
- Научно-практический клинический центр диагностики и телемедицинских технологий
- Городская клиническая онкологическая больница № 1
- Московский государственный медико-стоматологический университет имени А.И. Евдокимова
- Выпуск: Том 2, № 3 (2021)
- Страницы: 301-312
- Раздел: Систематические обзоры
- URL: https://journals.rcsi.science/DD/article/view/70306
- DOI: https://doi.org/10.17816/DD70306
- ID: 70306
Цитировать
Аннотация
Цель ― оценка возможности метода магнитно-резонансной томографии (МРТ) органов грудной клетки для выявления лёгочных узлов, подозрительных в отношении злокачественности, в сравнении с компьютерной томографией (КТ).
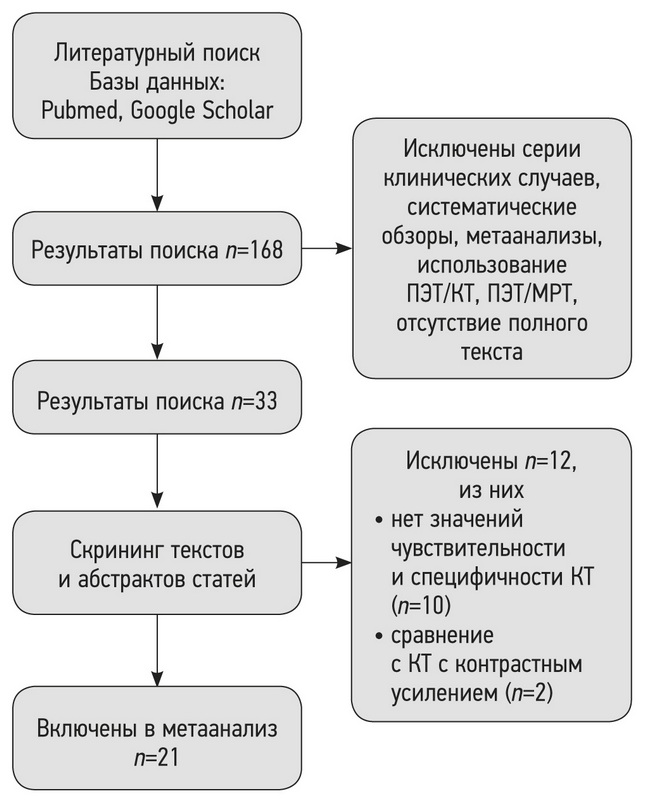
Материалы и методы. Проведён поиск в базах данных PubMed и Google Scholar за период до 7 апреля 2021 г. включительно. В соответствии с критериями соответствия были отобраны исследования, в которых проводилась оценка способности МРТ и КТ к выявлению лёгочных узлов, подозрительных в отношении злокачественности. Выбор метода анализа и группировки данных о чувствительности и специфичности выполняли по результатам оценки гетерогенности исследований. Для оценки статистической гетерогенности исследований, включённых в метаанализ, применяли критерий согласия Пирсона χ2 и индекс гетерогенности I2.
Результаты. По результатам поиска было отобрано 168 работ, в метаанализ вошло 21 исследование. Отобранные работы включали 1188 пациентов. По результатам метаанализа выявлено наличие статистически значимой гетерогенности p <0,00001 по критерию χ2 и индекс гетерогенности I2=99% для чувствительности и специфичности. В связи с этим для анализа данных использовали метод случайных эффектов. Значения чувствительности для МРТ находились в диапазоне от 70,4 до 100%, специфичности ― от 60,6 до 100%.
Заключение. МРТ обладает достаточной чувствительностью и специфичностью для определения злокачественности лёгочных узлов, обнаруженных при КТ-диагностике.
Ключевые слова
Полный текст
Открыть статью на сайте журналаОб авторах
Юрий Александрович Васильев
Научно-практический клинический центр диагностики и телемедицинских технологий; Городская клиническая онкологическая больница № 1
Email: dr.vasilev@me.com
ORCID iD: 0000-0002-0208-5218
SPIN-код: 4458-5608
кандидат медицинских наук
Россия, 127051, Москва, ул. Петровка, д. 24, стр. 1; МоскваОльга Юрьевна Панина
Научно-практический клинический центр диагностики и телемедицинских технологий; Городская клиническая онкологическая больница № 1; Московский государственный медико-стоматологический университет имени А.И. Евдокимова
Email: o.panina@npcmr.ru
ORCID iD: 0000-0002-8684-775X
SPIN-код: 5504-8136
Scopus Author ID: 57219837311
мл. научный сотрудник
Россия, 127051, Москва, ул. Петровка, д. 24, стр. 1; Москва; 127473, Москва, ул. Делегатская, д. 20, стр. 1Евгения Андреевна Грик
Московский государственный медико-стоматологический университет имени А.И. Евдокимова
Email: evgeniyagrik@gmail.com
ORCID iD: 0000-0002-7908-3982
SPIN-код: 5558-7307
MD
Россия, 127473, Москва, ул. Делегатская, д. 20, стр. 1Екатерина Сергеевна Ахмад
Научно-практический клинический центр диагностики и телемедицинских технологий
Email: e.ahmad@npcmr.ru
ORCID iD: 0000-0002-8235-9361
SPIN-код: 5891-4384
Россия, 127051, Москва, ул. Петровка, д.24, стр.1
Юлия Николаевна Васильева
Московский государственный медико-стоматологический университет имени А.И. Евдокимова
Автор, ответственный за переписку.
Email: drugya@yandex.ru
ORCID iD: 0000-0003-4955-2749
SPIN-код: 9777-2067
кандидат медицинских наук
Россия, 127473, Москва, ул. Делегатская, д. 20, стр. 1Список литературы
- Ost D., Fein A.M., Feinsilver S.H. Clinical practice. The solitary pulmonary nodule//N Engl J Med. 2003. Vol. 348, N 25. Р. 2535–2542. doi: 10.1056/NEJMcp012290
- Nasim F., Ost D.E. Management of the solitary pulmonary nodule//Curr Opin Pulm Med. 2019. Vol. 25, N 4. Р. 344–353. doi: 10.1097/MCP.0000000000000586
- MacMahon H., Naidich D.P., Goo J.M., et al. Guidelines for management of incidental pulmonary nodules detected on CT images: From the Fleischner Society 2017//Radiology. 2017. Vol. 284, N 1. Р. 228–243. doi: 10.1148/radiol.2017161659
- Liberati A., Altman D.G., Tetzlaff J., et al. The PRISMA statement for reporting systematic reviews and meta-analyses of studies that evaluate health care interventions: Explanation and elaboration//PLoS Med. 2009. Vol. 6, N 7. Р. e1000100. doi: 10.1371/journal.pmed.1000100
- Whiting P.F., Rutjes A.W., Westwood M.E., et al. Quadas-2: A revised tool for the quality assessment of diagnostic accuracy studies//Ann Intern Med. 2011. Vol. 155, N 8. Р. 529–536. doi: 10.7326/0003-4819-155-8-201110180-00009
- Higgins J.P., Thomas J., Chandler J., et al. Cochrane handbook for systematic reviews of interventions. John Wiley & Sons, Hoboken; 2019. doi: 10.1002/9781119536604
- Both M., Schultze J., Reuter M., et al. Fast T1- and T2-weighted pulmonary MR-imaging in patients with bronchial carcinoma//Eur J Radiol. 2005. Vol. 53, N 3. P. 478–488. doi: 10.1016/j.ejrad.2004.05.007
- Bruegel M., Gaa J., Woertler K., et al. MRI of the lung: Value of different turbo spin-echo, single-shot turbo spin-echo, and 3D gradient-echo pulse sequences for the detection of pulmonary metastases//J Magn Reson Imaging. 2007. Vol. 25, N 1. P. 73–81. doi: 10.1002/jmri.20824
- Meier-Schroers M., Kukuk G., Homsi R., et al. MRI of the lung using the PROPELLER technique: Artifact reduction, better image quality and improved nodule detection//Eur J Radiol. 2016. Vol. 85, N 4. P. 707–713. doi: 10.1016/j.ejrad.2015.12.016
- Meier-Schroers M., Homsi R., Schild H.H., Thomas D. Lung cancer screening with MRI: characterization of nodules with different non-enhanced MRI sequences//Acta Radiol. 2019. Vol. 60, N 2. P. 168–176. doi: 10.1177/0284185118778870
- Ohno Y., Koyama H., Yoshikawa T., et al. Standard-, reduced-, and nodose thin-section radiologic examinations: Comparison of capability for nodule detection and nodule type assessment in patients suspected of having pulmonary nodules//Radiology. 2017. Vol. 284, N 2. P. 562–573. doi: 10.1148/radiol.2017161037
- Regier M., Schwarz D., Henes F.O., et al. Diffusion-weighted MR-imaging for the detection of pulmonary nodules at 1.5 Tesla: Intraindividual comparison with multidetector computed tomography//J Med Imaging Radiat Oncol. 2011. Vol. 55, N 3. P. 266–274. doi: 10.1111/j.1754-9485.2011.02263.x
- Satoh S., Kitazume Y., Ohdama S., et al. Can malignant and benign pulmonary nodules be differentiated with diffusion-weighted MRI?//Am J Roentgenol. 2008. Vol. 191, N 2. P. 464–470. doi: 10.2214/AJR.07.3133
- Schaefer J.F., Schneider V., Vollmar J., et al. Solitary pulmonary nodules: Association between signal characteristics in dynamic contrast enhanced MRI and tumor angiogenesis//Lung Cancer. 2006. Vol. 53, N 1. P. 39–49. doi: 10.1016/j.lungcan.2006.03.010
- Schroeder T., Ruehm S.G., Debatin J.F., et al. Detection of pulmonary nodules using a 2D HASTE MR sequence: comparison with MDCT//Am J Roentgenol. 2005. Vol. 185, N 4. P. 979–984. doi: 10.2214/AJR.04.0814
- Sommer G., Tremper J., Koenigkam-Santos M., et al. Lung nodule detection in a high-risk population: Comparison of magnetic resonance imaging and low-dose computed tomography//Eur J Radiol. 2014. Vol. 83, N 3. P. 600–605. doi: 10.1016/j.ejrad.2013.11.012
- Vogt F.M., Herborn C.U., Hunold P., et al. HASTE MRI versus chest radiography in the detection of pulmonary nodules: comparison with MDCT//Am J Roentgenol. 2004. Vol. 183, N 1. P. 71–78. doi: 10.2214/ajr.183.1.1830071
- Yi C.A., Jeon T.Y., Lee K.S., et al. 3-T MRI: usefulness for evaluating primary lung cancer and small nodules in lobes not containing primary tumors//Am J Roentgenol. 2007. Vol. 189, N 2. P. 386–392. doi: 10.2214/AJR.07.2082
- Chang S., Hong S.R., Kim Y.J., et al. Usefulness of thin-section single-shot turbo spin echo with half-fourier acquisition in evaluation of local invasion of lung cancer//J Magn Reson Imaging. 2015. Vol. 41, N 3. P. 747–754. doi: 10.1002/jmri.24587
- Schaefer J.F., Vollmar J., Schick F., et al. Solitary pulmonary nodules: Dynamic contrast-enhanced MR imaging — Perfusion differences in malignant and benign lesions//Radiology. 2004. Vol. 232, N 2. P. 544–553. doi: 10.1148/radiol.2322030515
- Kono R., Fujimoto K., Terasaki H., et al. Dynamic MRI of solitary pulmonary nodules: comparison of enhancement patterns of malignant and benign small peripheral lung lesions//Am J Roentgenol. 2007. Vol. 188, N 1. P. 26–36. doi: 10.2214/AJR.05.1446
- Feng H., Shi G., Liu H., et al. Free-breathing radial volumetric interpolated breath-hold examination sequence and dynamic contrast-enhanced MRI combined with diffusion-weighted imaging for assessment of solitary pulmonary nodules//Magn Reson Imaging. 2021. Vol. 75. P. 100–106. doi: 10.1016/j.mri.2020.10.009
- Kim J.H., Kim H.J., Lee K.H., et al. Solitary pulmonary nodules: A comparative study evaluated with contrast-enhanced dynamic MR imaging and CT//J Comput Assist Tomogr. 2004. Vol. 28, N 6. P. 766–775. doi: 10.1097/00004728-200411000-00007
- Ohno Y., Nishio M., Koyama H., et al. Solitary pulmonary nodules: Comparison of dynamic first-pass contrast-enhanced perfusion area-detector CT, dynamic first-pass contrast-enhanced MR imaging, and FDG PET/CT//Radiology. 2015. Vol. 274, N 2. P. 563–575. doi: 10.1148/radiol.14132289
- Heye T., Sommer G., Miedinger D., et al. Ultrafast 3D balanced steady-state free precession MRI of the lung: Assessment of anatomic details in comparison to low-dose CT//J Magn Reson Imaging. 2015. Vol. 42, N 3. P. 602–609. doi: 10.1002/jmri.24836
- Akata S., Kajiwara N., Park J., et al. Evaluation of chest wall invasion by lung cancer using respiratory dynamic MRI//J Med Imaging Radiat Oncol. 2008. Vol. 52, N 1. P. 36–39. doi: 10.1111/j.1440-1673.2007.01908.x
- Hittmair K., Eckersberger F., Klepetko W., et al. Evaluation of solitary pulmonary nodules with dynamic contrast-enhanced MR imaging-a promising technique?//Magn Reson Imaging. 1995. Vol. 13, N 7. P. 923–933. doi: 10.1016/0730-725x(95)02010-q
- Alper F., Kurt A.T., Aydin Y., et al. The role of dynamic magnetic resonance imaging in the evaluation of pulmonary nodules and masses//Med Princ Pract. 2013. Vol. 22, N 1. P. 80–86. doi: 10.1159/000339475
- Frericks B.B., Meyer B.C., Martus P., et al. MRI of the thorax during whole-body MRI: Evaluation of different MR sequences and comparison to thoracic multidetector computed tomography (MDCT)//J Magn Reson Imaging. 2008. Vol. 27, N 3. P. 538–545. doi: 10.1002/jmri.21218
- Cieszanowski A., Lisowska A., Dabrowska M., et al. MR imaging of pulmonary nodules: Detection rate and accuracy of size estimation in comparison to computed tomography//PLoS One. 2016. Vol. 11, N 6. P. e0156272. doi: 10.1371/journal.pone.0156272
- Ohno Y., Hatabu H., Takenaka D., et al. Solitary pulmonary nodules: Potential role of dynamic MR imaging in management — Initial experience//Radiology. 2002. Vol. 224, N 2. P. 503–511. doi: 10.1148/radiol.2242010992
- Zou Y., Zhang M., Wang Q., et al. Quantitative investigation of solitary pulmonary nodules: dynamic contrast-enhanced MRI and histopathologic analysis//Am J Roentgenol. 2008. Vol. 191, N 1. P. 252–259. doi: 10.2214/AJR.07.2284
- Dewes P., Frellesen C., Al-Butmeh F., et al. Comparative evaluation of non-contrast CAIPIRINHA-VIBE 3T-MRI and multidetector CT for detection of pulmonary nodules: In vivo evaluation of diagnostic accuracy and image quality//Eur J Radiol. 2016. Vol. 85, N 1. P. 193–198. doi: 10.1016/j.ejrad.2015.11.020
- Fatihoğlu E., Biri S., Aydın S., et al. MRI in evaluation of solitary pulmonary nodules//Turkish Thorac J. 2019. Vol. 20, N 2. P. 90–96. doi: 10.5152/TurkThoracJ.2018.18049
- Heye T., Ley S., Heussel C.P., et al. Detection and size of pulmonary lesions: How accurate is MRI? A prospective comparison of CT and MRI//Acta Radiol. 2012. Vol. 53, N 2. P. 153–160. doi: 10.1258/ar.2011.110445
- Koo C.W., Lu A., Takahashi E.A., et al. Can MRI contribute to pulmonary nodule analysis?//J Magn Reson Imaging. 2019. Vol. 49, N 7. P. e256–e264. doi: 10.1002/jmri.26587
- Koyama H., Ohno Y., Kono A., et al. Quantitative and qualitative assessment of non-contrast-enhanced pulmonary MR imaging for management of pulmonary nodules in 161 subjects//Eur Radiol. 2008. Vol. 18, N 10. P. 2120–2131. doi: 10.1007/s00330-008-1001-2
- Koyama H., Ohno Y., Seki S., et al. Value of diffusion-weighted MR imaging using various parameters for assessment and characterization of solitary pulmonary nodules//Eur J Radiol. 2015. Vol. 84, N 3. P. 509–515. doi: 10.1016/j.ejrad.2014.11.024
- Huang Y.S., Niisato E., Su M.Y., et al. Detecting small pulmonary nodules with spiral ultrashort echo time sequences in 1.5 T MRI//MAGMA. 2021. Vol. 34, N 3. P. 399–409. doi: 10.1007/s10334-020-00885-x
- Ying G.S., Maguire M.G., Glynn R.J., et al. Calculating sensitivity, specificity, and predictive values for correlated eye data//Investig Ophthalmol Vis Sci. 2020. Vol. 61, N 11. P. 29. doi: 10.1167/iovs.61.11.29
- Bradley S.H., Kennedy M.P., Neal R.D. Recognising lung cancer in primary care//Adv Ther. 2019. Vol. 36, N 1. P. 19–30. doi: 10.1007/s12325-018-0843-5
- Nikolаev E., Gombolevskiy V, Gonchar AP, et al. Incidental findings during lung cancer screening by low-dose computed tomography//Tuberc Lung Dis. 2018. Vol. 96, N 11. P. 60–67. doi: 10.21292/2075-1230-2018-96-11-60-67
- Loverdos K., Fotiadis A., Kontogianni C., et al. Lung nodules: A comprehensive review on current approach and management//Ann Thorac Med. 2019. Vol. 14, N 4. P. 226–238. doi: 10.4103/atm.ATM_110_19